what is a hydrogen bond? group of answer choices a special type of dipole-dipole attraction involving hydrogen bound to another hydrogen atom a special type of attraction involving any molecules that contain hydrogens a special type of dipole-dipole attraction involving hydrogen bound to any other atom none of the above a special type of dipole-dipole attraction involving hydrogen bound to a highly electronegative atom
Answers
Instead of forming a covalent bond with an atom of hydrogen, hydrogen bonds are a specific form of dipole-dipole attraction between molecules.
Briefing:
It results from the attractive force between a hydrogen atom covalently bound to an extremely electronegative atom, like an N, O, or F atom, and an additional extremely electronegative atom.
The partial positive charge of the hydrogen atoms in some polar molecules that contain hydrogen atoms are drawn to the partial negative charge of an atom in another molecule close by. It is known as a hydrogen bond to describe this attraction. Only three elements on the current periodic table, oxygen, nitrogen, and fluorine, can create hydrogen bonds.
To know more about periodic table, visit:
https://brainly.com/question/11155928
#SPJ4
Related Questions
the tendency of a mineral to break and produce smooth, curving, shell-shaped surfaces is termed:
Answers
The tendency of a mineral to break and produce smooth, curving, shell-shaped surfaces is termed "conchoidal fracture." This characteristic is observed in certain minerals due to their specific atomic structure and bonding patterns. Minerals with this property exhibit a predictable breakage pattern, creating distinctive curving surfaces that resemble the shape of a seashell or a conch.
Conchoidal fracture is commonly seen in minerals with strong covalent or ionic bonds, such as quartz and obsidian. These minerals lack distinct cleavage planes, so when they break, they tend to form these smooth, curved surfaces. The absence of cleavage planes is a result of the uniform distribution of bonds throughout the mineral, which causes them to fracture in a more random manner, creating the curving shape.
In summary, the term "conchoidal fracture" refers to the tendency of a mineral to break and produce smooth, curving, shell-shaped surfaces. This property is observed in minerals with strong covalent or ionic bonds and a lack of distinct cleavage planes, such as quartz and obsidian. The unique breakage pattern is due to the uniform distribution of bonds within the mineral's structure, resulting in a random, curved fracture pattern.
Learn more about Conchoidal fracture here: brainly.com/question/30444209
#SPJ11
Rank the following carbocations in order of decreasing stability.
most stable
least stable
[C+]C(=C)C1CCCCC1
[C+]C1CCCCC1
C[C+]1CCCCC1
Answers
Answer:
english
Explanation:
language rank the following carbocation in orde of dicrising stability
The correct order of decreasing stability of the carbocations is 1, 3, 2.
The order of stability of carbocations is given as: Tertiary> Secondary> Primary> Methyl. Tertiary carbocations are the most stable because they have three methyl groups that release electrons.
A carbocation is a molecule containing a carbon atom with a positive charge and 3 bonds. Basically, it's a carbon cation. It used to be called carbonium ion.
CnH2n + x is the carbocation formula. The carbocation formula can have as many carbons as it wants, but not more than one hydrogen. It can also have as many hydrogens as it wants, as long as it has at least one carbon.
To learn more about carbocations, refer to the link:
https://brainly.com/question/29750400
#SPJ6
a student prepared an agno3 (aq) stock solution by dissolving an unknown mass of agno3 in a 250.00 ml volumetric flask and filling to the line with water. the student added 25.059 ml of the stock solution to a 500 ml volumetric flask and filled to the line with water to make 500 ml of a 9.443x10-3 m agno3 solution? what mass (in g) of agno3(s) did the student add to the 250 ml volumetric flask to make the stock solution?
Answers
Using the molarity of the final solution and the molar mass of silver nitrate, we can calculate that the student used 8.0015 g of silver nitrate to make the stock solution.
First, we calculate the number of moles in the 500.00 mL flask using the molarity (c) of the final solution and its volume (V = 500.00 mL = 0.50000 L).
c = n/V ⇒ n = c*V
n = 9.443 * 10⁻³ M * 0.50000 L
n = 4.7215 * 10⁻³ mol
This same number of moles was in the 25.059 mL solution that was subsequently diluted to 500.00 mL. Knowing this, we can make a simple proportion to calculate the number of moles present in the original 250.00 mL:
4.7215 * 10⁻³ mol : 25.059 mL = X : 250.00 mL
X = 4.7215 * 10⁻³ mol * 250.00 mL / 25.059 mL
X = 0.047104 mol
That's the amount (n) of silver nitrate used to make the stock solution. Now we can convert it to mass (m) by using the molar mass of silver nitrate (M = 169.87 g/mol):
n = m/M ⇒ m = n*M
m = 0.047104 mol * 169.87 g/mol
m = 8.0015 g
You can learn more about molarity here:
brainly.com/question/2817451
#SPJ4
According to the solubility curves below - if you have a solution with 60g KNO₃ in 100mL at 20C of water what would you classify the solution as?
a. Unsaturated
b. Saturated
c. Supersaturated
d. Would not make a solution
(I missed this lesson please explain)
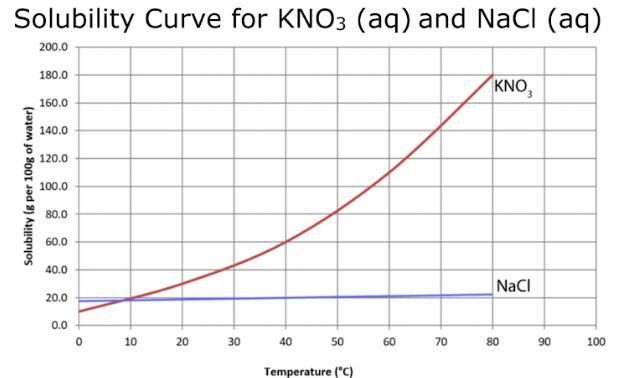
Answers
The correct classification for the given solution is: a. Unsaturated. Option A
Based on the provided solubility curve, which shows the relationship between temperature and solubility of KNO₃ in water, we can determine the classification of the solution with 60g KNO₃ in 100mL of water at 20°C.
From the solubility curve, we can see that the solubility of KNO₃ in water at 20°C is approximately 100g/100mL. Since the solution contains 60g of KNO₃, which is less than the maximum solubility of 100g at 20°C, we can classify the solution as unsaturated.
An unsaturated solution means that it has not reached its maximum solute concentration at a given temperature and can dissolve more solute. In this case, the solution with 60g KNO₃ in 100mL of water at 20°C still has the capacity to dissolve more KNO₃.
Therefore, the correct classification for the given solution is: a. Unsaturated.
It is important to note that solubility can vary with temperature, so the classification may change if the temperature of the solution is altered. Option A.
For more such questions on Unsaturated visit:
https://brainly.com/question/530542
#SPJ8
Calculate the value of ΔG∘rxnΔGrxn∘ for hypochlorous acid at 296 K. Ka = 2.9 × 10–8 and assume Ka does not change significantly with temperature. $$HClO(aq)+H2O(l) HClO−(aq)+H3O+(aq)
Answers
The standard Gibbs free energy change (ΔG∘rxn) for the reaction involving hypochlorous acid (HClO) at 296K is determined using ΔG∘rxn =-RTln(K), where R is the gas constant, T is the temperature in K.
To calculate ΔG∘rxn, we use the equation ΔG∘rxn = -RTln(K), where R is the gas constant (8.314 J/(mol·K)) and T is the temperature in Kelvin. In this case, the equilibrium constant (K) is determined by the acid dissociation constant (Ka) for HClO. The equilibrium constant expression for the reaction is K = [HClO−][H3O+]/[HClO].
Given that Ka = 2.9 × 10^–8, we substitute this value into the K expression. Next, we calculate ΔG∘rxn using the given temperature of 296 K. By plugging in the values into the equation ΔG∘rxn = -(8.314 J/(mol·K))(296 K)ln(2.9 × 10^–8) and evaluating the expression, we can determine the value of ΔG∘rxn for the reaction involving HClO at 296 K.
To learn more about acid, click here: brainly.com/question/31333804
#SPJ11
when 0.367 mol of a weak acid, hx, is dissolved in 2.00 l of aqueous solution, the ph of the resultant solution is 2.60. calculate ka for hx. report your answer rounded to two significant figures using e- notation.
Answers
when 0.367 mol of a weak acid, hx, is dissolved in 2.00 l of aqueous solution, the ph of the resultant solution is 2.60. ka for hx is 3.405 × \(10^{-5}\)
Number of moles = 0.367
Volume of solution = 2l
concentration = 0.367/2 = 0.1835 mol/L
ph = 2.60
we know
ph = - log [H+]
2.51 × \(10^{-2.60}\)M = [H+]
The acid HX dissociate as
HX → H+ + X-
The acid dissociation constant Ka, for the dissociation reaction is
Ka = [H+][X-]/[HX] ; at equilibrium, [H+] = [X-]
Ka = 3.405 × \(10^{-5}\)
A solution in which water serves as the solvent is called an aqueous solution. The most common way to represent it in chemical equations is to add (aq) to the appropriate chemical formula. For instance, the formula for a solution of table salt, or sodium chloride (NaCl), in water is Na+(aq) + Cl The word aqueous, which derives from the word aqua, means that something is connected to, resembles, or is dissolved in water. Water is a common solvent in chemistry because it is an excellent solvent and abundant in nature. Since water is frequently used as the experiment's solvent, unless otherwise stated, the term "solution" refers to an aqueous solution.
Learn more about Aqueous solution here:
https://brainly.com/question/14097392
#SPJ4
What is the coordination number of the central metal in [Cr(CO) 6
] ? The CO ligand can be classified as: What is the coordination number of the central metal in Na 3
[CuCl 5
]? The Cl −
ligand can be classified as:
Answers
The coordination number of the central metal in [Cr(CO)6] is 6. This means that there are 6 ligands attached to the central chromium atom. The CO ligand is classified as a monodentate ligand, meaning it can donate one electron pair to the central metal.
In the case of Na3[CuCl5], the coordination number of the central metal is 5. This means that there are 5 ligands attached to the central copper atom. The Cl- ligand is classified as a monodentate ligand, meaning it can donate one electron pair to the central metal.
To summarize, the coordination number for [Cr(CO)6] is 6, and the coordination number for Na3[CuCl5] is 5.
To know more about coordination number, visit at:
https://brainly.com/question/27289242
#SPJ11
Calculate the number of grams of 4.9 % (m/m) NaCl solution that contains 7.10 g of NaCl Express your answer to two significant figures and include the appropriate units.
Answers
The mass of the 4.9% (m/m) NaCl solution that contains 7.10 g of NaCl is 145 g.
How many grams of the 4.9% (m/m) NaCl solution contains 7.10 g of NaCl?
In order to calculate the mass of the NaCl solution, we need to consider the concentration of the solution, which is given as 4.9% (m/m). This means that there are 4.9 grams of NaCl for every 100 grams of the solution.
To find the mass of the NaCl solution, we can set up a proportion based on the given information:
(4.9 g NaCl / 100 g solution) = (7.10 g NaCl / x g solution)
Cross-multiplying and solving for x, we can calculate the mass of the solution:
x = (7.10 g NaCl) * (100 g solution) / (4.9 g NaCl)x ≈ 145 gTherefore, approximately 145 grams of the 4.9% (m/m) NaCl solution contain 7.10 g of NaCl.
Learn more about mass
brainly.com/question/11954533
#SPJ11
A lump of zinc is tossed into a beaker of 500L of 14M hydrochloric acid. this reaction produces Hydrogen Gas and zinc (II) chloride. If the hydrogen gas is combusted and produces 645L of water vapor at 400 kelvin and 1.75 atm, what is the mass of the zinc?
Answers
If the hydrogen gas is combusted and produces 645L of water vapor at 400 kelvin and 1.75 atm, 2796.96 g mass of the zinc is produced .
Using the ideal gas law equation:
PV = nRT
n = (PV) / (RT)
= (1.75 atm * 645 L) / (0.0821 atm·L/(mol·K) * 400 K)
= 42.71 moles
the balanced equation for the reaction between zinc and hydrochloric acid:
Zn + 2HCl -> \(ZnCl_{2}\) + \(H_{2}\)
1 mole of zinc produces 1 mole of hydrogen gas. Therefore, the moles of zinc are also 42.71.
The molar mass of zinc is 65.38 g/mol.
Mass of zinc = moles of zinc * molar mass of zinc
= 42.71 moles * 65.38 g/mol
= 2796.96 g
Therefore, the mass of the zinc is 2796.96 grams.
learn more about hydrogen gas :
https://brainly.com/question/30829657
Question 3 (3 points)
(03.04LO)
Match the formula to the correct name. (3 points)
Column A
Column B
1.
Cas
a. Calcium sulfide
2.
CaSO4
b. Calcium sulfate
3.
CaSO3
C. Calcium sulfite
Answers
Answer:
Column A \({}\) Column B
1. CaS \({}\) Calcium Sulfide
2. CaSO₄ \({}\) Calcium sulfate
3. CaSO₃ \({}\) Calcium sulfite
Explanation:
1) Calcium sulfide is a compound formed between calcium, Ca, and sulfur, S, with a chemical formula of CaS. It forms cubic crystals similar to rock salt, and smells like rotten eggs
2) Calcium sulfate is a chemical compound consisting of calcium, Ca, and sulfate SO₄. It is found in nature in the hydrated form as the mineral gypsum
3)Calcium sulfite is a chemical compound formed between calcium, Ca and sulfite, SO₃ used to treat water and preserve food.
Answer:
the formula for calcium sulfite is CaSO3
the formula for calcium sulfate is CaSO4
the formula for calcium sulfide is CaS
Explanation:
because of our belief in the law of conservation of mass, it seems reasonable that
Answers
Because of our belief in the law of conservation of mass, it seems reasonable that matter cannot be created or destroyed, only changed in form or transferred from one substance to another.
This law is fundamental to our understanding of the physical world, and it has many practical applications in fields like chemistry, physics, and engineering. It helps us to predict the outcomes of chemical reactions, to balance equations, and to understand the behavior of materials under different conditions.
The law of conservation of mass is a fundamental principle that underlies much of what we know about the world around us, and it will continue to play an important role in scientific research and development for years to come.
More on law of conservation of mass: https://brainly.com/question/28711001
#SPJ11
A 10.0 g piece of hot metal at 300. °C was dropped into a 150.0 g sample of cooler room temperature water that was initially 25.0 °C. If 6.25 kJ of heat was transferred, what was the final temperature of the water?What was the specific heat of the metal?
Answers
Answer:
The final temperature of water is 34.92 ⁰C
The specific heat of the metal is 2357.78 J/kg⁰C
Explanation:
Given;
mass of the hot metal, m = 10.0 g = 0.01 kg
temperature of the hot metal, \(t_m\) = 300 °C
mass of water, \(m_w\) = 150 g = 0.15 kg
initial temperature of the water, \(t_w_i\) = 25.0 °C
heat lost by the hot metal = heat gained by water
Q = 6.25 kJ = 6250 J
let the final temperature of water = T
\(Q = m_w C_p_w (T-t_w_i)\\\\T-t_w_i = \frac{Q}{m_wC_p_w}\\\\ T= \frac{Q}{m_wC_p_w} + t_w_i\\\\T = \frac{6250}{(0.15)(4200)} + 25\\\\T = 34.92 ^0 C\)
The final temperature of water is also the equilibrium temperature.
The specific heat of the metal is given by
\(Q = mC_p_m (300 - T)\\\\C_p_m = \frac{Q}{m(300 - T)}\\\\ C_p_m = \frac{6250}{0.01(300 - 34.92)}\\\\ C_p_m = 2357.78 \ J/kg ^0C\)
if 5-ml of a 5m solution of naoh is placed in a 250-ml volumetric flask and diluted to the mark with water, what would be the concentration of the diluted solution?
Answers
If 5-ml of a 5m solution of naoh is placed in a 250-ml volumetric flask and diluted to the mark with water, then the concentration of the diluted solution is 0.1M.
What is Concentration?Concentration is a term which is defined as the ratio of number of moles of compound or substance to the volume of the substance in litre.
Dilution is inversely proportional to the concentration of the solution. It can be expressed as
M1V1 = M2V2
where,
M1 is the initial concentration
M2 is the final concentration
V1 is the initial volume
V2 is the final volume
Given,
M1 = 5M
V1 = 5 ml
V2 = 250 ml
By substituting all the values, we get
5 × 5 = M2 × 250
M2 = 25 / 250
M2 = 0.1 M
Thus, we concluded that If 5-ml of a 5m solution of naoh is placed in a 250-ml volumetric flask and diluted to the mark with water, then the concentration of the diluted solution is 0.1M.
learn more about concentration:
https://brainly.com/question/13494737
#SPJ4
pls help AHHHHHHHHHHHH

Answers
Answer:
1 Zn + 2 HCl ⇒ 1 H₂ + 1 ZnCl₂
Hope this helps!!!
will give brainliest for the quickest and correct answer
An atom X has 5 energy levels of electrons outside its nucleus and 74 neutrons inside its nucleus. Identify this atom
a. I
b. Cd
c. Ca
d. Fr
e. P
Answers
Answer:
Iodine
Explanation:
Iodine is located in the 5th period, meaning that it will have 5 energy levels.
Additionally, Iodine has an atomic mass of about 127 atomic mass units, and since it has 53 protons, it will have 74 neutrons.
Let me know if this helps!
Explanation:
e is right now answer for teaching us
Which of the following does NOT regulate flux through the citric acid cycle?
A.) Inhibition of α-ketoglutarate dehydrogenase by succinyl CoA
B.) Allosteric inhibition of isocitrate dehydrogenase by ADP
C.) Ca2+ activation of isocitrate dehydrogenase
D.) The ratio of [NAD+]/[NADH]
E.) Concentration of intermediates
Answers
The answer is E.) Concentration of intermediates. The citric acid cycle, also known as the Krebs cycle or the tricarboxylic acid (TCA) cycle, is a central metabolic pathway that takes place in the mitochondria of eukaryotic cells.
It plays a crucial role in the oxidative metabolism of carbohydrates, fats, and amino acids, generating high-energy electron carriers (NADH and FADH2) and producing ATP through oxidative phosphorylation. The regulation of flux through the citric acid cycle is important for maintaining energy homeostasis and adapting to metabolic demands. Several factors influence the rate of the citric acid cycle, and most of them are involved in feedback mechanisms that ensure proper coordination with other metabolic pathways. However, the concentration of intermediates is not a direct regulatory factor.
Inhibition of α-ketoglutarate dehydrogenase by succinyl CoA: Succinyl CoA is an intermediate in the citric acid cycle and acts as a negative regulator of α-ketoglutarate dehydrogenase, the enzyme responsible for catalyzing the conversion of α-ketoglutarate to succinyl CoA. This feedback inhibition helps regulate the rate of the cycle and prevents the buildup of excessive intermediates. while factors such as the inhibition of α-ketoglutarate dehydrogenase by succinyl CoA, allosteric inhibition of isocitrate dehydrogenase by ADP, and Ca2+ activation of isocitrate dehydrogenase play important roles in regulating flux through the citric acid cycle.
To learn more about citric acid cycle click here; brainly.com/question/30392607
#SPJ11
which of the statements is/are true about batteries? i. the overall reaction in a battery must have a positive emf. ii. batteries must have some sort of salt bridge to allow for ion transport. iii. lithium ion batteries provide higher voltages than most other batteries. which of the statements is/are true about batteries? i. the overall reaction in a battery must have a positive emf. ii. batteries must have some sort of salt bridge to allow for ion transport. iii. lithium ion batteries provide higher voltages than most other batteries. only iii is true. only i is true. all of i, ii, and iii are true. only ii is true. only ii and iii are true.
Answers
Statement i is true about batteries. For a battery to be able to supply electrical energy to a circuit, the overall reaction that occurs within the battery must have a positive emf.
i. The overall reaction in a battery must have a positive emf is true. The emf, or electromotive force, is the measure of the energy per unit charge that is supplied by a source of electrical energy, such as a battery.
ii. Batteries must have some sort of salt bridge to allow for ion transport is partially true. A salt bridge is a component of some types of batteries that allows for the transport of ions between the two half-cells of the battery. However, not all batteries require a salt bridge. For example, some types of batteries, such as the alkaline battery, use a porous separator instead of a salt bridge to allow for ion transport electrical energy .
iii. Lithium-ion batteries provide higher voltages than most other batteries is partially true. Lithium-ion batteries are known for their high energy density and long cycle life, but their voltage is not necessarily higher than other types of batteries. The voltage of a battery depends on the chemistry of the reaction that occurs within the battery, and different types of batteries can have different voltages.
Learn more about electrical energy here
https://brainly.com/question/863273
#SPJ11
Help me thank you....

Answers
Answer:3
Explanation:
it is b
Answer:
The correct answer is . . . "B" .
♡ ( i just got the answer from that other dude- ) ♡
Explanation:
Give 5 examples of branches of science, then give an example of situations that apply the use of it in daily activities. Example: PHYSICS - Turning the light on and off.
Answers
Answer:
See explanation
Explanation:
Biology- digestion of food materials
Chemistry- dissolving salt in water
Astronomy - observing day and night
Physics - watching a television
Geography - observing weather changes during the day
Can someone please check my answer for question 2? If It’s wrong please correct me. Thank you

Answers
The new volume : V₂ = 1185.9 cm³
Further explanationGiven
V₁ = 571 cm³
T₁ = 260 K
T₂ = 540 K
Required
The new volume (V₂)
Solution
Charles's Law
When the gas pressure is kept constant, the gas volume is proportional to the temperature
\(\tt \dfrac{V_1}{T_1}=\dfrac{V_2}{T_2}\)
Input the value :
V₂ = V₁.T₂/T₁
V₂ = 571 x 540 / 260
V₂ = 1185.9 cm³
Answer:
V₁ = 571 cm³
T₁ = 260 K
T₂ = 540 K your right
Explanation:
The new volume will by 1185.9
What is the mass of an object with a density of 5.7 g/cm^3 and a volume of 48
cm^3? *
Answers
Answer:
273.6 g
Explanation:
mass =(density)(volume)
mass=(5.7g/cm^3 )(48 cm^3)
cancel out your cm^3 and your are left with grams
multiply (5.7g)(48)=273.6 g
The mass of an object with a density of 5.7 g/cm³ and a volume of 48 cm³ is 273.6 grams.
What is density?Density is defined as the degree to which a material is packed together.
It is also defined as a mass per unit volume of the substance.
Its SI unit is kilogram per cubic meter or gram per meter.
Density can be expressed as
Density = mass / volume
Mass = density x volume
As given, Density = 5.7 g/cm³
Volume = 48 cm³
So, Mass = 5.7 g/cm³ x 48 cm³
= 273.6 grams
Thus, the mass of an object with a density of 5.7 g/cm³ and a volume of 48 cm³ is 273.6 grams.
To learn more about density, refer to the link below:
https://brainly.com/question/15164682
#SPJ5
What is the volume in liters that is needed to create a 0.5 M solution with 4.9 moles of Fe(OH)2? Round your answer to 1 decimal.
Answers
Answer:
\(0.5\: moles\: occupies \: 1 \: litre \\ 4.9 \: moles \: will \: occupy \: ( \frac{4.9}{0.5} ) \: moles \\ = 9.8 \: litres\)
PLZ help, I'm really behind in school! I will mark brainliest for correct answers!
If the distance between two objects is reduced by a factor of 5, how will Fg be affected?
A. F g decreases to 1/25
B. F g increases by 5 times
C. F g decreases to 1/5
D. F g increases by 25 times
Answers
Answer:
Gravitational force is given by:
F=G×
d
2
m×M
Distance between two masses is increased s.t. new distance is D= 5 d
New gravitational force F
1
=F
Let on of the mass is changed to m
1
so as to maintain the same gravitational force.
Answer:
D
Explanation:
F g increases by 25 times
A very large tank initially contains 100 L of pure water. Starting at time t=0 a solution with a salt concentration of 0.3 kg/L is added at a rate of 7 L/min. The solution is kept thoroughly mixed and is drained from the tank at a rate of 5 L/min. Answer the following questions. 1. Let y(t) be the amount of salt (in kilograms) in the tank after t minutes. What differential equation does y satisfy? Use the variable y for y(t). Answer (in kilograms per minute):
dt/dy = 2. How much salt is in the tank after 40 minutes? Answer (in kilograms):
Answers
1. The differential equation satisfied by y(t) is: dy/dt = 0.6 kg/min
The amount of salt in the tank after t minutes can be represented by the function y(t). We need to find the differential equation that y satisfies.
Initially, the tank contains 100 L of pure water, which means there is no salt in the tank. As time passes, a solution with a salt concentration of 0.3 kg/L is added at a rate of 7 L/min. The salt concentration in the tank will increase with the addition of this solution.
At the same time, the solution is drained from the tank at a rate of 5 L/min. This will result in a decrease in the salt concentration in the tank.
To find the differential equation satisfied by y(t), we need to consider the rate of change of salt in the tank.
Rate of change of salt in the tank = Rate of salt added - Rate of salt drained
The rate of salt added is given by the product of the concentration of the solution (0.3 kg/L) and the rate at which the solution is added (7 L/min). So, the rate of salt added = 0.3 kg/L * 7 L/min.
The rate of salt drained is given by the product of the concentration of the solution (0.3 kg/L) and the rate at which the solution is drained (5 L/min). So, the rate of salt drained = 0.3 kg/L * 5 L/min.
Therefore, the differential equation satisfied by y(t) is:
dy/dt = (0.3 kg/L * 7 L/min) - (0.3 kg/L * 5 L/min)
Simplifying the equation:
dy/dt = 2.1 kg/min - 1.5 kg/min
dy/dt = 0.6 kg/min
So, the differential equation satisfied by y(t) is:
dy/dt = 0.6 kg/min
2. The amount of salt in the tank after 40 minutes is 24 kilograms.
To find the amount of salt in the tank after 40 minutes, we can solve the differential equation.
dy/dt = 0.6 kg/min
Integrating both sides with respect to t:
∫dy = ∫0.6 dt
Integrating, we get:
y = 0.6t + C
To find the value of C, we can use the initial condition that the tank initially contains 100 L of pure water, which means there is no salt. So, at t = 0, y = 0.
Substituting these values into the equation:
0 = 0.6(0) + C
C = 0
Therefore, the equation becomes:
y = 0.6t
Now, we can find the amount of salt in the tank after 40 minutes by substituting t = 40 into the equation:
y = 0.6(40)
y = 24 kg
Learn more about differential equations from the given link:
https://brainly.com/question/1164377
#SPJ11
The volume of a sample of water is found to be 86.3 cubic centimeters (cm3). What is the volume of the sample in mm3?
Answers
answer; 8.017*10^3
Explanation: because cube means it is multiplication of three times so we multiply 86.3 three times and again we multiply it with 100 because we need to convert it in mm and at last we put the answer in cube root ..
what is lower explosive limit?
Answers
The lower explosive limit (LEL) is the lowest concentration of a combustible gas or vapor in air at which it can ignite.
The lower explosive limit is typically expressed as a percentage of the lower flammability limit (LFL). For example, methane has an LEL of 5% and an LFL of 4.4%, meaning that the lower explosive limit for methane is 5% of 4.4%. This means that if the concentration of methane in air is between 4.4% and 5%, then it is considered to be at its lower explosive limit.It is incorrect to equate the percentage measurement on flammable air detectors with the LFL concentrations.
learn more about methane Refer:brainly.com/question/28933327
#SPJ4
Which of the following compounds is (are) a conjugated diene(s)? A. 4-methyl-1,3-heptadience B. 3-methyl-1,5-heptadience C. 2-methyl-2,4-heptadience D. 4-methyl-1,4-heptadience E. 5-methyl-2,3-heptadience
Answers
Out of the compounds given, A, B, D, and E are conjugated dienes because they contain a chain of alternating double bonds.
In a conjugated diene, the double bonds are separated by a single bond, which results in a system of overlapping p-orbitals that form a delocalized pi-electron system. This delocalization stabilizes the molecule and can lead to unique chemical reactivity.
Compound C, on the other hand, is not a conjugated diene because it has two adjacent double bonds, which means there is no single bond separating them. Therefore, the p-orbitals of the double bonds cannot overlap and form a delocalized pi-electron system.
In summary, A, B, D, and E are conjugated dienes, while C is not. It is important to note the differences between these compounds' structures and understand how they affect the properties and behavior of the compounds.
To know more about conjugated dienes visit:
https://brainly.com/question/30755456
#SPJ11
A, B, C, or D, thanks for the help

Answers
According to the label the true statement about the following substance is option c. It does not pose a health risk
What are lab labels ?
Specially designed labels with particularly specialised adhesive qualities are called "lab labels." In the pharmaceutical or medical industry, as well as in laboratories, they are used for labelling. For the labelling of tubes, test tubes, blood bags, and other types of vessels, special laboratory labels are created.
Labels on chemicals
The identify of the hazardous chemical(s) in the container (for example, chemical name) and the dangers present must both be listed on this label. This warning about hazards can be distributed in a variety of ways. Employers want to choose a solution that will function in each place.
The following label indicates that the chemical does not pose a health risk.
Learn more about lab labels here https://brainly.com/question/14459928
#SPJ10
a. calculate δh for the forward reaction b. calculate δs for the forward reaction c. calculate δssurr for the forward reaction d. calculate δssurr for the reverse reaction
Answers
The ΔH for the forward reaction, you need to subtract the enthalpy of the reactants from the enthalpy of the products. ΔH = ΣH(products) - ΣH(reactants). Enthalpy (H) represents the heat content of a system. For example, if the enthalpy of the products is 100 kJ/mol and the enthalpy of the reactants is 50 kJ/mol, then ΔH = 100 kJ/mol - 50 kJ/mol = 50 kJ/mol. ΔH represents the heat released or absorbed during a chemical reaction.
b. To calculate ΔS for the forward reaction, you need to subtract the entropy of the reactants from the entropy of the products. ΔS = ΣS(products) - ΣS(reactants). Entropy (S) represents the degree of disorder in a system. For example, if the entropy of the products is 200 J/mol·K and the entropy of the reactants is 100 J/mol·K, then ΔS = 200 J/mol·K - 100 J/mol·K = 100 J/mol·K. ΔS represents the change in disorder during a chemical reaction.
c. ΔSsurr represents the change in entropy of the surroundings. It can be calculated using the equation ΔSsurr = -ΔH/T, where T is the temperature in Kelvin. ΔSsurr shows how the surroundings (not the system) change during a reaction. For example, if ΔH is -100 kJ/mol and T is 300 K, then ΔSsurr = -100 kJ/mol / 300 K = -0.33 kJ/(mol·K).
d. ΔSsurr for the reverse reaction can be calculated using the same equation as in c, but with the opposite sign for ΔH. So if ΔH for the reverse reaction is +100 kJ/mol and T is 300 K, then ΔSsurr = +100 kJ/mol / 300 K = +0.33 kJ/(mol·K).
To know more about enthalpy visit:-
https://brainly.com/question/32882904
#SPJ11
Pls describe the water system in the southwest USA and current complications. describe two or three ways that everyone can conserve water.
Answers
Answer:
It has become evident in recent years that established water policies, under the current laws and management regimes that have allowed a flourishing agriculture and economy in the Southwest, are not appropriate to the challenges facing water demand in the 21st century. Yet only limited institutional and policy steps have been taken to develop new tools and approaches addressing these challenges. The circumstances listed below illustrate some of the more important unsustainable aspects of water use in the Southwest.