Answers
Answer:
B i think
Explanation:
Related Questions
Name 2 physical properties that would allow differentiation between copper, lead and iron shot.
Answers
Answer:
Two physical properties that would allow differentiation between copper, lead and iron shot are - Color and Density
COPPER - Color - Copper has a reddish brown color .
Density - Density of copper is 8.96 \(g/cm^{3}\)
LEAD - Color - Lead is grey in color.
Density - Density of Lead is 11.34\(g/cm^{3}\)
IRON SHOT - Color- Iron shot is in grey color with extra shine .
Density - Density of iron shot is 10.49 \(g/cm^{3}\)
Explanation:
COPPER- Copper (Cu) is a reddish, highly ductile metal that belongs to Group 11 (Ib) of the periodic table and is a particularly excellent conductor of electricity and heat. Copper is found in nature in its free metallic state.
LEAD- Lead (Pb) is a soft, silvery white or grayish metal in Periodic Group 14 (IVa). Lead is a weak conductor of electricity and is very malleable, ductile, and thick. Lead, which has been known since antiquity and is considered the oldest of metals by alchemists, is extremely durable and corrosion-resistant, as evidenced by the continued use of lead water pipes constructed by the ancient Romans.
IRON SHOT - Small round iron balls used as projectiles are known as iron shot. Balance scales are used with simple weights. Iron balls are used in the shot put competition.
Hence , the differentiation is mentioned in the above answer.
even one or two crystals of copper sulphate can make its solution in water coloured blue. why
Answers
this also may happen because of the water molecules that get attached
If a jar of saltwater sits in the sun, the liquid level steadily decreases, and finally, crystals appear. Explain what is happening.
Answers
When a jar containing saline solution is exposed to sunlight, its liquid content gradually diminishes and eventually forms crystals as a result of the evaporation process.
What is the saltwater about?The energy from the sun's heat accelerates the movement of molecules in saltwater, which leads to their subsequent release from the liquid surface as water vapor. As a result of this process, the moisture transforms into gas and ascends, scattering throughout the atmosphere.
The concentration of salt in the remaining liquid increases as water evaporates from saltwater, as salt is unable to evaporate like water. As time passes, the salt concentration within the water triples to a point where it becomes incapable of remaining dissolved and commences the process of crystal formation.
Learn more about saltwater from
https://brainly.com/question/18761619
#SPJ1
an atom of which element has the strongest attraction for electons in a chemical bond?cholorine, phosphorus, carbon, sulfur
Answers
The number of atoms of any element in the given chemical formula is the number that is written on the foot of the symbol of that element. Therefore, chlorine has the strongest attraction for electrons in a chemical bond.
What is atom?Atom is the smallest particle of any matter. Atom combines to form element and element combine to form molecule or compound.
Atom consists of electron, proton and neutron. The total mass of atom is inside the nucleus. Inside the nucleus proton and neutron is there. So calculate mass of an atom, total mass of all protons is added to the total mass of neutron. Electrons revolve around the nucleus.
Electronegative element have greatest strongest attraction for electrons due to high effective nuclear charge. Among the given molecules, chlorine is the most electronegative element. Chlorine has the strongest attraction for electrons in a chemical bond.
Therefore, chlorine has the strongest attraction for electrons in a chemical bond.
Learn more about atoms, here:
https://brainly.com/question/13518322
#SPJ2
Animal fats and vegetable oils are triacylglycerols, or triesters, formed from the reaction
of glycerol (1, 2, 3-propanetriol) with three long-chain fatty acids. One of the methods
used to characterize a fat or an oil is a determination of its saponification number. When
treated with boiling aqueous KOH, an ester is saponified into the parent alcohol and fatty
acids (as carboxylate ions). The saponification number is the number of milligrams of
KOH required to saponify 1.000 g of the fat or oil. In a typical analysis, a 2.085-g sample
of butter is added to 25.00 mL of 0.5131 M KOH. After saponification is complete, the
excess KOH is back titrated with 10.26 mL of 0.5000 M HCl. What is the saponification
number for this sample of butter?
Answers
Saponification number = (V × M × F × 56.1) / W
Where:
V = volume of HCl used in the back titration
M = molarity of HCl
F = factor of KOH (which is 1 for pure KOH)
W = weight of the butter sample used in grams
First, we need to calculate the amount of KOH used in the saponification reaction:
0.5131 M KOH = 0.5131 moles KOH / liter
25.00 mL KOH = 0.02500 L KOH
moles KOH used = 0.5131 moles/L × 0.02500 L = 0.0128 moles KOH
Since the saponification reaction is a 1:1 reaction between KOH and the triacylglycerol in the butter sample, the amount of butter used is also 0.0128 moles.
Next, we need to calculate the amount of HCl that reacted with the excess KOH:
0.5000 M HCl = 0.5000 moles HCl / liter
10.26 mL HCl = 0.01026 L HCl
moles HCl used = 0.5000 moles/L × 0.01026 L = 0.00513 moles HCl
Since the reaction between HCl and KOH is also a 1:1 reaction, the moles of KOH that were not used in the saponification reaction is equal to the moles of HCl used in the back titration:
moles KOH not used = moles HCl used = 0.00513 moles HCl
To find the saponification number,
Saponification number = (V × M × F × 56.1) / W
Saponification number = (0.01026 L × 0.5000 moles/L × 1 × 56.1) / 2.085 g
Saponification number = 6.50
Therefore, the saponification number for this sample of butter is 6.50.
To know more about Saponification:
https://brainly.com/question/2263502
#SPJ1
1. How is energy being used inefficiently?
2.What should you do to make it efficient?
pls answer thank you :)
Answers
Answer:
1. Energy intensity (primary energy consumption divided by GDP) for the world and various countries. The lower the value, the less energy is used to generate economic output, so lower means more efficient in a sense.
2.Lower Your Thermostat. Adopt the habit of lowering the temperature on your thermostat while away from home.
Start a Compost Pile.
Install Low-Flow Showerheads.
Seal All Windows.
Limit Space Heater Use.
Turn Off Unnecessary Water.
Replace Incandescent Bulbs.
Unplug Unused Chargers.
Give TWO reasons, in terms of shielding, why potassium is the
most reactive of the three elements.
Answers
Answer:
Hi..? All three elements are in group 1 thus they have 1 valence electron which is easy to lose due to shielding.As Pottasium is larger than the rest,Pottasium's valence electron is at a greater distance from the attractive nucleus and is so removed more easily than Sodium's and Lithium's valence electrons.As it is removed more easily it requires less energy and can be said to be more reactive
A pure copper cube has an edge length of 3.00 cm
How many copper atoms does it contain? (volume of a cube = (edge length)^3
; density of copper = 8.96 g/cm^3)

Answers
Since this equals (edge length)3, the cube's volume is 3 * 1.12 cm, or 3.36 cm3. As a result, the cube has 3.36/8.96 g/cm3 = 0.43 grammes of copper atoms.
What is the perimeter of a volumetric cube?V= s3, where s is the length of one of the cube's edges, is the formula for a cube's volume. The length of the edge is equal to the volume's cube root, or 4 cm, if the volume is 64.
How do I figure out the atom count?To get the number of moles in an element or compound, divide the supplied mass by the element or compound's molar mass.
To know more about copper visit:-
brainly.com/question/26474177
#SPJ1
True or False, a skeleton equation shows the formulas of the compounds participating in a chemical reaction and their states of matter but not the ratio in which they react.
Answers
The given statement is true. A skeleton equation shows the formulas of the compounds participating in a chemical reaction and their states of matter but not the ratio in which they react.
What does a skeleton equation show?A skeleton equation defines the chemical formulas and physical states of the reactants and products, yet it does not account for the relative amounts of the reactants and products. Chemical reactions result simply from the rearrangement of atoms, thus the law of conservation of mass have to be observed.
Skeleton Equations are the equations where there is only chemical formula of reactants and products but no state mentioned and no balancing of atoms on either side of the equation done. For example: Mg + O2 → MgO, it is a skeleton equation. In the equation, the only thing mentioned is the chemical formula of reactant.
Learn more skeleton equation at: https://brainly.com/question/19909826
#SPJ4
prop-1-yne + 2HBr/H2O2 = A;
A + 2H2O = B;
B + K2CO3(aq) = C;
C + heat = D;
D + HBr = E.
find the compounds A, B, C, D and E
Answers
Based on the given reactions, the compounds are as follows:
A: The specific product formed from the reaction between prop-1-yne and either 2HBr or H2O2.
B: The product formed when compound A reacts with 2H2O.
C: The product formed when compound B reacts with K2CO3(aq).
D: The product formed from the heat-induced reaction of compound C.
E: The product formed when compound D reacts with HBr.
Based on the given reactions, let's analyze the compounds involved:
Reaction 1: prop-1-yne + 2HBr/H2O2 = A
The reactant prop-1-yne reacts with either 2HBr or H2O2 to form compound A. The specific product formed will depend on the reaction conditions.
Reaction 2: A + 2H2O = B
Compound A reacts with 2H2O (water) to form compound B.
Reaction 3: B + K2CO3(aq) = C
Compound B reacts with K2CO3(aq) (potassium carbonate dissolved in water) to form compound C.
Reaction 4: C + heat = D
Compound C undergoes a heat-induced reaction to form compound D.
Reaction 5: D + HBr = E
Compound D reacts with HBr (hydrobromic acid) to form compound E.
For more such questions on compounds
https://brainly.com/question/704297
#SPJ8
d = ? v = 100 ml. m = 1500 g
Answers
Answer:
15
Explanation:
Determine if each statement is an acid, base, or neutral.- removes OH- ions from water- removes H+ ions from water
Answers
Answer
An acid removes OH⁻ ions from water.
A base removes H⁺ ions from water.
Explanation
An acid is a chemical species that donates protons or hydrogen ions and/or accepts electrons. Most acids contain a hydrogen atom bonded that can release (dissociate) to yield a cation and an anion in water.
Hence, an acid removes OH- ions from water.
A base is a substance that results in an increase in the concentration of hydroxide ions when it is dissolved in water. Also, a base is a substance that when dissolved in water increases the number of hydroxide ions.
Therefore, a base removes H+ ions from water.
I NEED THIS RIGHT NOW!! Daria had some sand from the beach. The mass of the sand was 72 grams. She used the graduated cylinder below to measure the volume.
What is the volume of the sand found in the graduated cylinder? _____ mL

Answers
Daria had some beach sand with her. The sand has a 72 gramme mass. She calculated the volume using the graduated cylinder below. The graduated cylinder contains 15 mL of sand.
The volume of the sand is calculated using the graduated cylinder below. The sand's bulk is specified as 72 grammes.
We can use the water displacement method to calculate the volume of the sand. Following is a description of how to estimate the amount of sand using the water displacement method:
The graduated cylinder of water should first be measured for volume.
The graduated cylinder's water volume should then be measured after adding the sand to it. The volume of water increases by the same amount.
Let's use the provided problem to implement this approach.
In the beginning, there is 10 mL of water in the graduated cylinder. The graduated cylinder contains 25 mL of water once the sand has been added.
The amount of sand is therefore equal to the difference between the two volumes, which is: Sand volume equals final water volume minus initial water volume (25 - 10 = 15 mL).
As a result, there are 15 mL of sand in the graduated cylinder.
Answer : 15
For more such questions on volume, click on:
https://brainly.com/question/14197390
#SPJ8
Photoelectric effect will occur only if frequency of light striking an electron in a metal is above a certain threshold frequenci
Answers
The statement is correct. The photoelectric effect refers to the phenomenon where electrons are ejected from the surface of a material when it is exposed to light. The frequency of light striking an electron in a metal must be above the threshold frequency in order for the photoelectric effect to occur.
The statement is correct. The photoelectric effect refers to the phenomenon where electrons are ejected from the surface of a material when it is exposed to light. However, for the photoelectric effect to occur, the frequency of the incident light must be above a certain threshold frequency.
The threshold frequency is the minimum frequency of light required to dislodge electrons from the material. Below this threshold frequency, regardless of the intensity or duration of the light, no electrons will be emitted.
This behavior can be explained by the particle-like nature of light, where light is composed of discrete packets of energy called photons. The energy of a photon is directly proportional to its frequency. Only photons with energy greater than or equal to the binding energy of the electrons in the material can dislodge them.
Therefore, the frequency of light striking an electron in a metal must be above the threshold frequency in order for the photoelectric effect to occur.
For more question on photoelectric
https://brainly.com/question/1458544
#SPJ8
Draw the structure of phosphatidylserine and discuss its components
Answers
Phosphatidylserine is a type of phospholipid that is mainly found in cell membranes. Its structure is made up of two fatty acid chains, a phosphate group, a serine molecule, and a glycerol molecule.
The fatty acid chains are hydrophobic, meaning they repel water, while the phosphate group and serine molecule are hydrophilic, meaning they attract water.
The glycerol molecule acts as a bridge that connects the two fatty acid chains to the phosphate group and serine molecule.
The structure of phosphatidylserine is important for its function in the cell membrane.
Because of the hydrophobic and hydrophilic components of its structure, phosphatidylserine is able to form a lipid bilayer, which is a barrier that separates the inside of the cell from the outside environment.
The hydrophilic heads of the phosphatidylserine molecules face outward and interact with water, while the hydrophobic tails face inward and repel water.
Phosphatidylserine also plays a role in cell signaling and apoptosis, which is programmed cell death.
It acts as a signaling molecule by binding to proteins that are involved in cellular pathways.
In addition, phosphatidylserine is translocated to the outer leaflet of the cell membrane during apoptosis, which signals to immune cells that the cell is ready to be removed.
In conclusion, the structure of phosphatidylserine is made up of two fatty acid chains, a phosphate group, a serine molecule, and a glycerol molecule. Its hydrophobic and hydrophilic components allow it to form a lipid bilayer in cell membranes, and it also plays a role in cell signaling and apoptosis.
For more such questions on Phosphatidylserine
https://brainly.com/question/16179573
#SPJ8
Please help!!
Is this an:
Acid
Base
Neither
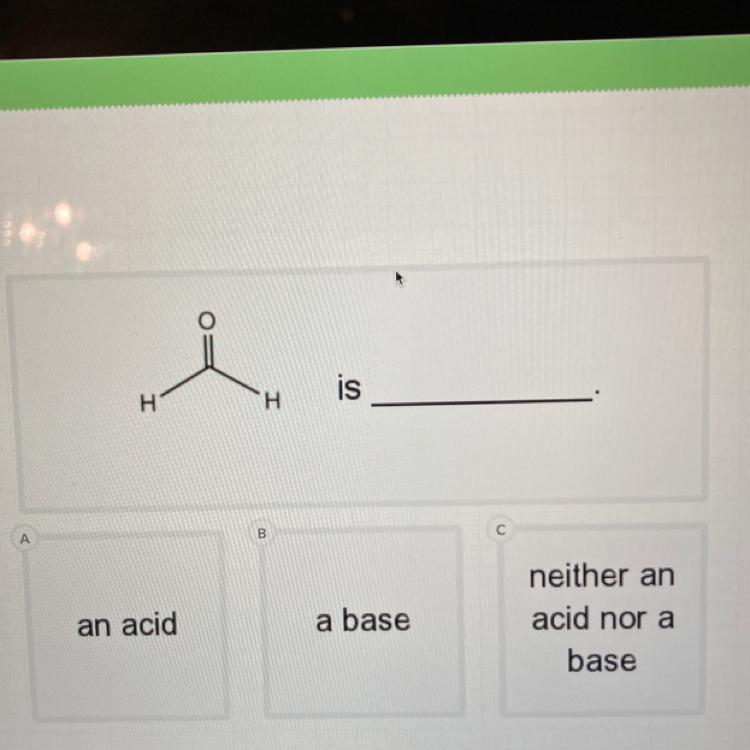
Answers
Answer:
it is a weak acid
acid
Explanation:
formaldehyde is a weak acid (pK greater than 13),
Some species of hares are brown most of the year, but change color to white in the winter. This allows them to
blend in through the seasons.
Where are these organisms adapted to live?
O deciduous forests
O desert
O tropical rain forests
O savanna
Answers
Answer:Deciduouse forest
Explanation:desert is too hot for snow, The rain forest always rains, Savannas are always warmer in climate.
Answer:
A
Explanation:
Identify the correct chemical formula. Select one: O a. K₂C₂H₂O2 0 b. K2(OH)2 O c. KCIO3 O d. 504 MATU 20 A www. wowow
Answers
Answer:
B.K2(OH)2 i think that is the answer
how do you think you can use lenses and mirrors to change the direction and intensity of a light wave?
Answers
How do elements cycle through the atmosphere?
Answers
The elements cycle through the atmosphere is biogeochemical cycle. In atmosphere different types of cycle are revolving around.
What is biogeochemical cycle?The biogeochemical cycle is refer to the exchange of substances between biotic and abiotic sources.
It can also be defined as the process by which a chemical material circulates through Earth's biotic and abiotic regions.
There are six types of biogeochemical cycle.
Water cycleCarbon cycleNitrogen cycleOxygen cyclePhosphorous cycleSulphur cycleThus, the elements cycle through the atmosphere is biogeochemical cycle. In atmosphere different types of cycle are revolving around.
To learn more about biogeochemical cycle, refer to the link below:
https://brainly.com/question/1204069
#SPJ1
Calculate the heat change (ΔΗ°rxn) for the slow reaction of zinc with water
Zn(s)+2H2O(l) ---> Zn^2+ (aq) +H2(g)
ΔΗ°rxn = kJ
Answers
The heat change or enthalpy change, ΔH°rxn, for the slow reaction of zinc with water is +417.7 kJ/mol.
The heat change or enthalpy change, ΔH°rxn, for the reaction of zinc (Zn) with water (H₂O) can be calculated using the standard enthalpies of formation for each species involved in the reaction.
Balanced chemical equation for the reaction is;
Zn(s) + 2H₂O(l) → Zn²⁺(aq) + H₂(g)
The standard enthalpy change for the reaction, ΔH°rxn, can be calculated as the sum of the standard enthalpies of formation of the products minus the sum of the standard enthalpies of formation of the reactants, each multiplied by their respective stoichiometric coefficients;
ΔH°rxn = Σ(nΔH°f, products) - Σ(mΔH°f, reactants)
where n and m are the stoichiometric coefficients of the products and reactants, respectively, and ΔH°f is the standard enthalpy of formation.
Assuming standard conditions (25°C and 1 atm), the standard enthalpies of formation for Zn²⁺(aq) and H₂(g) are typically tabulated values. Let's assume their values to be ΔH°f(Zn²⁺(aq)) = -153.9 kJ/mol and ΔH°f(H₂(g)) = 0 kJ/mol, respectively.
The standard enthalpy of formation of water (H₂O) is -285.8 kJ/mol.
Put the values into the equation, we get;
ΔH°rxn = [ΔH°f(Zn²⁺(aq)) + ΔH°f(H₂(g)] - [ΔH°f(Zn(s)) + 2ΔH°f(H₂O(l))]
ΔH°rxn = [-153.9 + 0] - [0 + 2(-285.8)]
ΔH°rxn = -153.9 + 571.6
ΔH°rxn = 417.7 kJ/mol
To know more about heat change here
https://brainly.com/question/18912282
#SPJ1
What is the empirical formula of C9H18
Answers
Answer:
CH2
Explanation:
there is a common factor for 9;18 which is 9 so u divide both numbers by it 9/9 and 18/9 giving 1:2 so 1 C to every 2 H so CH2
The empirical formula of C9H18 will be CH2.
What is the empirical formula?The empirical formula of a compound shows the ratio of atoms present in a compound. The molecular formula of the compound shows the total number of atoms present in a compound.
Here, C9H18 if we divide the number of total atoms by 9, C9/ 9, and H18/ 9. the ratio will be in the form of 1: 2. therefore, the empirical formula will be CH2.
C9H18 is the molecular formula of the compound, while CH2 is the empirical formula of the compound. The molecular formula of butane is C4H16, then its empirical formula is CH4. The molecular formula of compound is P4H10 then its empirical formula is P2H5.
Therefore, The empirical formula of C9H18 will be CH2.
To learn more about empirical formulas refer to the link:
https://brainly.com/question/14044066
#SPJ2
Write down the names of the elements

Answers
Answer:
Carbon and hydrogen
Explanation:
C = Carbon
H = Hydrogen
PLEASE NEEEED HELP drag each label to the correct location on the image here’s one way to follow the scientific method. Place the missing steps in the correct position in the process

Answers
Answer: Make an observation -> Ask a question. -> Construct a hypothesis. -> Test the hypothesis with an investigation -> Analyze the data -> Explain the results -> (Left) The hypothesis is true ------ (Right) The hypothesis is false. -> Communicate the results.
Explanation:
Make an observation -> Ask a question. -> Construct a hypothesis. -> Test the hypothesis with an investigation -> Analyze the data -> Explain the results -> (Left) The hypothesis is true ------ (Right) The hypothesis is false. -> Communicate the results.
Answer:
Make an observation -> Ask a question. -> Construct a hypothesis. -> Test the hypothesis with an investigation -> Analyze the data -> Explain the results -> (Left) The hypothesis is true ------ (Right) The hypothesis is false. -> Communicate the results.
Explanation:
Pls help !
What happens to the speed of a tsunami wave as it approaches the shore?
It increases and then decreases.
It increases.
It remains at a constant speed.
It decreases.
Which term represents when a tsunami wave slows down as it reaches shore?
surfing
waving
sinking
shoaling
Answers
Answer: It decreases.
Explanation:
How many electrons are being shared

Answers
In the carbon dioxide molecule has four shared pairs of electrons, there will be four covalent bonds created, hence option B is correct.
In organic chemistry, covalent bonds are far more prevalent than ionic ones. Two nuclei are simultaneously drawn to one or more pairs of electrons to form a covalent connection. Bonding electrons are those that are present between the two nuclei.
When atoms share electron pairs, covalent bonding results. Atoms create covalent bonds with one another in order to build a complete electron shell, which increases stability.
Learn more about covalent bonds, here:
https://brainly.com/question/19382448
#SPJ1
please help with this!

Answers
Answer:
d) The object falls towards the ground
Explanation:
, 2.2 g of Al reacts with 1.8 g of Oz.
Answers
The result of the reaction between 2.2 g of Al and 1.8 g of O2 is the formation of 3.89 g of \(Al_{2} O_{3}\).
The given masses of Al and \(O_{2}\) are in a ratio of approximately 1:1. Therefore, we can assume that all the Al reacts with all the O2, and use stoichiometry to determine the products and reactants involved.
The balanced chemical equation for the reaction between Al and O2 is:
4 Al + 3 \(O_{2}\) → 2 \(Al_{2} O_{3}\)
Using the molar masses of Al (26.98 g/mol) and \(O_{2}\) (32.00 g/mol), we can convert the given masses into moles:-
2.2 g Al × (1 mol Al/26.98 g Al) = 0.0815 mol Al
1.8 g \(O_{2}\) × (1 mol \(O_{2}\)/32.00 g \(O_{2}\)) = 0.0563 mol \(O_{2}\)
From the balanced chemical equation, we can see that 4 moles of Al react with 3 moles of \(O_{2}\) to produce 2 moles of Al2O3. Therefore, the limiting reactant is \(O_{2}\), since there are fewer moles of \(O_{2}\) than required by the stoichiometry of the reaction.
Using the mole ratio from the balanced chemical equation, we can determine the amount of \(Al_{2} O_{3}\) produced:
0.0563 mol \(O_{2}\) × (2 mol \(Al_{2} O_{3}\)/3 mol \(O_{2}\)) × (101.96 g Al2O3/mol) = 3.89 g \(Al_{2} O_{3}\).
To know more about chemical equation:
brainly.com/question/11231920
#SPJ1
(4 pts) Fill in the number of protons and electrons for each product and reactant (two boxes have been filled in for you). (2 pts) Verify that the number of protons on the left side of the chemical equation is equal to the number of protons of the right side. Show your work. (2 pts) Verify that the number of electrons on the left side of the chemical equation is equal to the number of electrons of the right side. Show your work. (3 pts) Which substance is being oxidized
Answers
Answer:
a)
Zn(s) + 2 H⁺(aq) ⇒ Zn²⁺(aq) + H₂(g)
#p⁺ 30 2 30 2
#e⁻ 30 0 28 2
b) 32
c) 30
d) Zn
Explanation:
There is some info missing. I will add the complete question.
Consider the following oxidation/reduction reaction.
Zn(s) + 2 H⁺(aq) ⇒ Zn²⁺(aq) + H₂(g)
#p⁺ 2
#e⁻ 2
a) Fill in the number of protons and electrons for each product and reactant (two boxes have been filled in for you).
b) Verify that the number of protons on the left side of the chemical equation is equal to the number of protons of the right side. Show your work.
c) Verify that the number of electrons on the left side of the chemical equation is equal to the number of electrons of the right side. Show your work.
d) Which substance is being oxidized?
a) The atomic number of Zn is 30 so it will have 30 protons. Since it ts neutral, it will also have 30 electrons. Zn²⁺ will also have 30 protons but it lost 2 electrons, so it has 28 electrons. The atomic number of H⁺ is 1, so each H atom will have 1 proton (2 in total). But since H has 1 electron, and H⁺ lost 1 electron, H⁺ will have 0 electrons. The complete chart is:
Zn(s) + 2 H⁺(aq) ⇒ Zn²⁺(aq) + H₂(g)
#p⁺ 30 2 30 2
#e⁻ 30 0 28 2
b) The total number of protons on the left side is: 30 + 2 = 32.
The total number of protons on the right side is: 30 + 2 = 32.
c) The total number of electrons on the left side is: 30 + 0 = 30.
The total number of electrons on the right side is: 28 + 2 = 30.
d) Zn(s) is oxidized because it loses electrons (from 30 to 28) and its number increases.
adjoa's gross salary for last month was ghs 4687.24. if ghs 421.78 was deducted from adjoa's salary pay,how much take home pay did adjoa earned this past month?
Answers
Since Ghs. 421.78 was deducted from Adjoa's salary pay, her take home pay is Ghs. 4265.46.
Given the following data:
Adjoa's gross salary = Ghs. 4687.24.Deduction = Ghs. 421.78Gross profit can be defined as the profit earned by a business after subtracting its cost of manufacturing and distributing goods from its total revenue (sales).
On a related note, gross salary is the amount of money earned by an individual before any deduction such as tax.
To find how much take home pay Adjoa earned this past month:
\(Take\;home = Gross\;salary - deduction\\\\Take\;home = 4687.24 - 421.78\)
Take home = Ghs. 4265.46.
Read more: https://brainly.com/question/25273589
