When using the pump dispenser, you should always change the volume dispensed to whatever you want.
True
False
Answers
The statement that one should always change the volume dispensed when using a pump dispenser is false, as the volume dispensed is typically pre-determined and cannot be changed, although adjustable settings may exist in some dispensers.
Can the volume dispensed from a pump dispenser be adjusted according to user preferences?Typically, the volume dispensed from a pump dispenser is pre-determined by its design and cannot be changed, although some dispensers may have adjustable settings that allow users to control the amount of product dispensed per pump.
What should be done if a pump dispenser has an adjustable setting for the volume dispensed, but it is dispensing too much or too little product per pump?If a pump dispenser has an adjustable setting, but is dispensing too much or too little product per pump, the user can adjust the setting to achieve the desired amount of product dispensed.
If the dispenser still does not dispense the desired amount after adjusting the setting, it may be faulty and require replacement.
To know more about Dispenser, visit:
https://brainly.com/question/13684628
#SPJ1
Related Questions
Calculate the relative formula mass of strontium nitrate, Sr(NO3)2.
(relative atomic masses: N = 14, O = 16, Sr = 88)
Answers
Answer:
its 210
Explanation:
Just add all atomic mass used in the formula together: 2*(14+16*3)+88= 2*62+88=124+88=210
Hope this was helpful
The relative formula mass of strontium nitrate Sr(NO₃)₂ is 210
The relative atomic masses of N, O and Sr are 14,16 and 88 respectively.
In calculating the relative atomic mass of an element with isotopes, the relative mass and proportion of each is taken into account. Adding the atomic masses together gives the relative formula mass of a compound
So, relative atomic mass of Sr(NO₃)₂ is calculated as
88+ 2(14+16×3) = 210
The atomic mass constant (symbol: mu) is defined as being 1/12 th of the mass of a carbon-12 atom. Since both quantities in the ratio are masses, the resulting value is dimensionless; hence the value is said to be relative atomic mass.
To know more about strontium nitrate here
https://brainly.com/question/26177156
#SPJ2
Suppose that some hydrochloric acid is placed in a beaker and a pH meter is set into the solution. It reads pH 1.5.
(a) Describe how the pH will change when a small amount of NaOH solution is added drop by drop to the acid. t
(b) Explain why the pH will change.
Answers
Answer:
A) The pH will increase.
B)The pH will change because the solution is being neutralized via a basic solution being added to an acid.
Ice at 0.0°C is mixed with 7.30 × 10^2 mL of water at 25.0°C. How much ice must melt to lower the water temperature to 0.0°C? The specific heat capacity of water is 4.186 J/(g·K). Latent heat of fusion for water is 333.7 J/g.
Answers
Approximately 35.90 grams of ice must melt to lower the water temperature to 0.0°C.
To solve this problem, we need to calculate the amount of heat that needs to be transferred from the water to the ice in order to lower the water temperature to 0.0°C.
First, let's calculate the initial heat content of the water. The specific heat capacity of water is 4.186 J/(g·K), and the mass of the water can be calculated using its density (1 g/mL) and volume (7.30 × 10^2 mL):
Mass of water = density × volume = 1 g/mL × 7.30 × 10^2 mL = 7.30 × 10^2 g
The initial heat content of the water can be calculated using the formula:
Heat content = mass × specific heat capacity × temperature change
Heat content = 7.30 × 10^2 g × 4.186 J/(g·K) × (25.0°C - 0.0°C) = 7.30 × 10^2 g × 4.186 J/(g·K) × 25.0°C
Next, we need to calculate the amount of heat that needs to be transferred from the water to the ice to lower the water temperature to 0.0°C. This heat transfer occurs during the melting of the ice.
The amount of heat required to melt the ice can be calculated using the formula:
Heat = mass of ice melted × latent heat of fusion
Let's assume that x grams of ice melts. The mass of the ice can be calculated using its density (0.92 g/mL) and volume (same as the volume of water):
Mass of ice = density × volume = 0.92 g/mL × 7.30 × 10^2 mL = 6.716 × 10^2 g
Heat = x g × 333.7 J/g
Now, we need to ensure that the heat transferred from the water to the ice is enough to lower the water temperature to 0.0°C. The heat transferred from the water to the ice is equal to the heat transferred from the water when its temperature drops to 0.0°C:
Heat content of water = Heat transferred to ice
7.30 × 10^2 g × 4.186 J/(g·K) × 25.0°C = x g × 333.7 J/g
Now, we can solve for x:
x = (7.30 × 10^2 g × 4.186 J/(g·K) × 25.0°C) / (333.7 J/g)
x ≈ 35.90 g
Therefore, approximately 35.90 grams of ice must melt to lower the water temperature to 0.0°C.
Learn more about temperature
https://brainly.com/question/27944554
#SPJ11
PLEASE HELP!!!!! BRAINLIEST WILL BE AWARDED (if correct)!!!
XeBr2Cl2 has 36 total valence electrons. What is the Lewis structure for XeBr2Cl2 that minimizes the formal charges on all atoms.
Answers
Answer:see below
Explanation:
:Cl: :Br:
|| ||
H3C-Xe-CH3
||
:Br:
Why is the use of high-frequency radio waves a beneficial technological advancement
for humans? Explain.
A. They have the best penetration.
B. They carry more information.
C. They travel over longer distances.
D. They do not require mediums to transmit.
I
Answers
Answer:
I think It's C
Explanation:
due to higher waves can get easy transfers and receive those signals for most things, such as radios, TVs, phone signals etc etc
The use of high-frequency radio waves, a beneficial technological advancement for humans because they travel over longer distances.
What is frequency ?The frequency of a repeated event is its number of instances per unit of time. In certain cases, it is also referred to as temporal frequency or ordinary frequency to underline differences with spatial and angular frequencies, respectively.
The number of waves that pass a specific place in a predetermined period of time is known as the wave frequency. The hertz (Hz) is the SI unit for wave frequency, where 1 hertz is equivalent to 1 wave crossing a fixed point in 1 second.
The vibrations that created the wave had the same frequency as the wave itself. For instance, you need to move a rope up and down faster to produce a wave with a greater frequency.
Thus, option C is correct.
To learn more about frequency, follow the link;
https://brainly.com/question/5102661
#SPJ2
What is the percent composition of each element in the compound below:
CrPO4
Answers
How many molecules are in 48.0 grams of NaOH?
Answers
Hope this helped :)
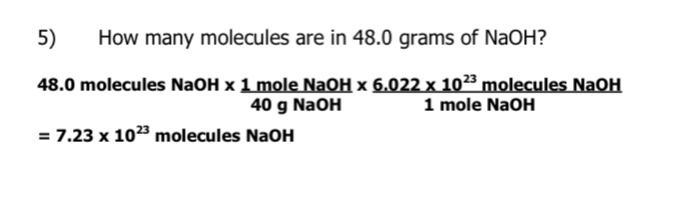
please answer this 1. rana wants to buy shirts for summer.should she buy cotton shirts or shirts made of synthetic material ? advise rana, give ur reason...
Answers
Answer:
Cotton Shirts
Explanation:
I would think that the cotton shirts would be more appropriate form of of clothing for summer.
Cotton has tiny pores that enable the sweat to evaporate off your skin, and hence keep you cooler. At the same time it absorbs this sweat, allowing easy evaporation. Synthetic fibers hold heat in, and do not allow sweat to evaporate.
It would be that Rana should buy cottons shirts for summer.
Question 14 PM2.5 is defined as ________
- the mass concentration of particles in the air less than or equal to 2.5 micrometers in diameter. - the mass concentration of particles in the air equal to 2.5 micrometers in diameter. - the mass concentration of particles in the air greater than or equal to 2.5 micrometers in diameter. Question 15 Carbon dioxide (CO2) is a criteria air pollutant. - True - False Question 16 Roughly percent of emissions of carbon monoxide in Santa Clara County come from mobile sources (select the choice closest to the correct answer). - 50 - 75 - 25 Question 17
The term "photochemical smog" is most synonymous with which of the following criteria air pollutants? - lead (Pb) - carbon monoxide (CO) - sulfur dioxide ( SO2) - ozone (O3) Question 18 "Attainment" of ambient air quality standards requires that measured concentrations at all monitoring stations within an air district are below ambient air standards. - True - False
Answers
: PM2.5 is defined as the mass concentration of particles in the air less than or equal to 2.5 micrometers in diameter.Question 15: False, carbon dioxide (CO2) is not considered a criteria air pollutant.
Question 16: The closest answer is 50%, but the exact percentage is not provided in the question.Question 17: The term "photochemical smog" is most synonymous with ozone (O3), which is a criteria air pollutant.Question 18: True, attainment of ambient air quality standards requires that measured concentrations at all monitoring stations within an air district are below ambient air standards.
Question 14 asks about the definition of PM2.5. PM2.5 refers to particulate matter with a diameter less than or equal to 2.5 micrometers. It represents the mass concentration of particles suspended in the air, which are small enough to be inhaled into the respiratory system and can have adverse health effects.
Question 15 states whether carbon dioxide (CO2) is a criteria air pollutant. Criteria air pollutants are a set of pollutants regulated by environmental agencies due to their detrimental impact on air quality and human health. However, carbon dioxide is not considered a criteria air pollutant because it does not directly cause harm to human health or the environment in the same way as pollutants like ozone or particulate matter.
Question 16 asks about the percentage of carbon monoxide (CO) emissions from mobile sources in Santa Clara County. While the exact percentage is not provided in the question, the closest answer option is 50%. However, it is important to note that the precise percentage may vary depending on specific local conditions and emissions sources.
Question 17 inquires about the criteria air pollutant most synonymous with the term "photochemical smog." Photochemical smog is primarily associated with high levels of ground-level ozone (O3). Ozone is formed when nitrogen oxides (NOx) and volatile organic compounds (VOCs) react in the presence of sunlight, creating a hazy and polluted atmospheric condition.
Question 18 addresses the concept of "attainment" of ambient air quality standards. To achieve attainment, measured concentrations of pollutants at all monitoring stations within an air district must be below the established ambient air quality standards. This ensures that the air quality in the given area meets the required standards for protecting human health and the environment.
Learn more about mass concentration here:- brainly.com/question/23437000
#SPJ11
Silicon has 2 isotopes, Silicon-28 with an abundance of 90% and Silicon-30 with an abundance of 10%. Find the AAM for Silicon.
Answers
Answer:
RAM
\(RAM = (mass \: of \: ^{28} Si \: \times \%abundance) + (mass \: of \: ^{30} Si \: \times \%abundance) \\ RAM = (28 \times 90\%) + (30 \times 10\%) \\ RAM = 25.2 + 3 \\ RAM = 28.2\)
1. list at least two possible sources of error from the experiment. would these errors result in an overestimation or an underestimation of the concentration of acetic acid in the vinegar
Answers
An error in estimation could arise owing to;
1) Inaccurate measurement of the volumes of the solutions
2) When the reaction vessels are not properly cleaned
What is error in an experiment?Error could occur in an experiment when values are misread. The error could be random error or a systematic error.
The major ways in which an error can occur in the estimation of concentration of acetic acid in the vinegar are;
1) Inaccurate measurement of the volumes of the solutions
2) When the reaction vessels are not properly cleaned
Learn more about errors in experiment:https://brainly.com/question/14149934
#SPJ1
Explain five physical and/or chemical properties that you need to consider for the substances before using them as construction materials for buildings
Answers
Answer: There are many properties of a metal which is taken into consideration when it is used for construction
Explanation:
The properties can be physical, chemical, et cetera. The physical properties that is considered are:
1. Durability: It can be defined as the property of the material to withstand the atmospheric pressure and other factors.
2. Density: The ratio of mass by volume of a material (homogeneous material) is called as density.
3. Fire resistance: The ability of the material to withstand in fire without changing its size or shape.
4. Weather resistance: The ability of the material to withstand the weather which could be variable round the year.
5.Water absorption: The ability of the material to absorb water and retain it.
The space around the nucleus of an atom that electrons orbit
Answers
Answer:
The electron cloud - or - electron orbitals
Explanation:
Protons and neutrons are held in the nucleus of the atom; electrons orbit around the nucleus in orbitals. Modern atomic theory tells us that these orbitals are not necessarily a circular or elliptical path but more like a cloud-like area where electrons are likely to be found at any given time.
HELP PLEASE❗️❗️‼️
An increase in temperature is an increase in.... ( there are two correct answers)
A.) Water molecules
B.) The melting point
C.) pressure
D.) Fun
Answers
Hope this helps!
which of the following are produced in the ozonolysis of the following molecule? the skeletal structure of a molecule with a smiles string of ccccCH3CH2COOH (CH3)2CHCOOH (CH3)2CHCH2COOH CH3CH2CH2COOH CH3CH2CH2CH2COOH
Answers
Propanedioic acid ((CH₃)₂C(O)COOH), oxalic acid (HO₂C-C(O)OH), and formic acid (HCOOH) are among the carboxylic acids created after the ozonolysis of ccccCH₃CH₂COOH.
What is ozonolysis?In an organic redox reaction known as ozonolysis, ozone is used to break unsaturated carbon-carbon bonds (double or triple bonds) in alkenes, alkynes, or azo compounds.
The molecule with the SMILES string ccccCH₃CH₂COOH can undergo ozonolysis to form a mixture of products. The ozonolysis reaction involves the cleavage of the carbon-carbon double bond by ozone (O₃) to form ozonide intermediates, which can then react further to form various products.
The ozonolysis of ccccCH₃CH₂COOH would result in the formation of several carboxylic acid products, including propanedioic acid ((CH₃)₂C(O)COOH), oxalic acid (HO₂C-C(O)OH), and formic acid (HCOOH). The exact ratio and amounts of these products depend on the specific conditions of the reaction, such as the concentration of ozone, temperature, and presence of any catalysts.
Therefore, the carboxylic acids produced in the ozonolysis of ccccCH₃CH₂COOH include propanedioic acid ((CH₃)₂C(O)COOH), oxalic acid (HO₂C-C(O)OH), and formic acid (HCOOH).
Learn more about ozonolysis on:
https://brainly.com/question/24113517
#SPJ11
Which option is a compound?
Answers
Examples of compounds include table salt or sodium chloride , sucrose (a molecule, nitrogen gas, a sample of copper, and water.
what is the role of hydrogen bonding in the ascent of xylem sap?
Answers
The role of hydrogen bonding in the ascent of xylem sap is quite complex and requires a long answer. Xylem sap is responsible for transporting water and nutrients from the roots to the leaves in plants, and it is transported through the xylem tissue.
The process of water transport in plants is primarily driven by a process called transpiration, which is the loss of water vapor from the leaves. As water evaporates from the leaves, it creates a negative pressure gradient that pulls water up from the roots. The water molecules are then pulled up through the xylem tissue due to the cohesive forces between water molecules, which is the attraction between molecules of the same substance.
Hydrogen bonding plays a key role in the cohesive forces between water molecules. The hydrogen atoms in one water molecule are attracted to the oxygen atoms in other water molecules, creating a network of hydrogen bonds that hold the water molecules together. This allows the water molecules to move in a continuous column, and as they move up the xylem tissue, they are able to pull more water molecules along with them due to the cohesive forces.
To know more about hydrogen bonding visit:-
https://brainly.com/question/31139478
#SPJ11
Nitrogen (N2) and hydrogen (H2) react to form ammonia (NH3). Consider a mixture of six nitrogen molecules and six hydrogen molecules in a closed container. Assuming the reaction goes to completion, what will the final product mixture be?
number of NH3 molecules
number of N2 molecules
number of H2 molecules
Answers
The balanced chemical equation for the reaction between nitrogen (\(N_{2}\)) and hydrogen (\(H_{2}\)) to form ammonia (\(NH_{3}\)) is:
\(N_{2}\) + 3\(H_{2}\) -> 2\(NH_{3}\)
In the given mixture of six nitrogen molecules and six hydrogen molecules, we have an equal number of both reactants. According to the stoichiometry of the reaction, one molecule of \(N_{2}\) reacts with three molecules of \(H_{2}\) to produce two molecules of \(NH_{3}\).
Since we have six molecules of each reactant, we can determine that three molecules of \(N_{2}\) will react with nine molecules of \(H_{2}\), resulting in six molecules of \(NH_{3}\) as the final product.
Therefore, the final product mixture will consist of six \(NH_{3}\) molecules, three \(N_{2}\) molecules (which did not react), and three \(H_{2}\) molecules (which did not react).
To know more about chemical equation, refer here:
https://brainly.com/question/28792948#
#SPJ11
Word Bank:
amount
atomic mass
atomic mass
atomic mass
atomic number
name
charge
electrons
neutrons
nucleus
protons
same
The number of protons in one atom of an element determines the atom’s _________________, and the number of electrons determines the _________________________ of the element.
The atomic number tells you the number of ___________________________ in one atom of an element. It also tells you the number of __________________________ in a neutral atom of that element. The atomic number gives the “identity” of an element as well as its location on the periodic table. No two different elements will have the ____________________ atomic number.
The ______________________ of an element is the average mass of an element’s naturally occurring atom, or isotopes, taking into account the ____________________ of each isotope.
The _______________________ of an element is the total number of protons and neutrons in the ___________________ of the atom.
The atomic mass is used to calculate the number of ______________________ in one atom of an element. In order to calculate the number of neutrons you must subtract the ______________________ from the ______________________.
Answers
Answer:
name,atomic mass,electrons,protons,same,charge,amount,atomic mass,charge,neutrons,atomic number,atomic mass
You were given a 100. G wine sample to verify its age. Using tritium dating you observe that the sample has 0. 688 decay events per minute. Tritium has a half life of 12. 3 and fresh water exhibits 5. 5 decay events per minute per 100g. What year was the wine produced?.
Answers
Wine was produced 37 years ago (1984 as usual year 15,2021) that is shown in the calculations below.
Reaction rate is calculated using the formula rate = Δ[C]/Δt, where Δ[C] is the change in product concentration during time period Δt. The rate of reaction can be observed by watching the disappearance of a reactant or the appearance of a product over time.
The time can be represented as follows:
t= 2.303\∧ log A0/A
∧= 0.693/t 1/2
The rate of a reaction is proportional to the reciprocal of the time taken. Rate α 1 time Rate is inversely proportional to time. Units: s-1, min-1 etc.
The given parameters are as follows:
t1/2=12.3
A0=5.5
A=0.688
t= 2.303/(0.693/12.3) log (5.5/0.688)
t=36.9
t=37 years
Thus, wine was produced 37 years ago (1984 as usual year 15,2021)
To learn more about rate of reaction check the link below:
https://brainly.com/question/24795637
#SPJ4
any atom is most stable when it’s outermost energy level contains
Answers
Explanation:
Atoms are at their most stable when their outermost energy level is either empty of electrons or filled with electrons. Sodium atoms have 11 electrons. Two of these are in the lowest energy level, eight are in the second energy level and then one electron is in the third energy level.
Any atom is most stable when it’s outermost energy level contains eight electrons.
What is an atom?An atom is defined as the smallest unit of matter which forms an element. Every form of matter whether solid,liquid , gas consists of atoms . Each atom has a nucleus which is composed of protons and neutrons and shells in which the electrons revolve.
The protons are positively charged and neutrons are neutral and hence the nucleus is positively charged. The electrons which revolve around the nucleus are negatively charged and hence the atom as a whole is neutral and stable due to presence of oppositely charged particles.
Atoms of the same element are similar as they have number of sub- atomic particles which on combination do not alter the chemical properties of the substances.
Learn more about atom,here:
https://brainly.com/question/13981855
#SPJ6
We would like to make a golden standard kilogram in the shape of circular cylinder. The density of gold is 19.32 g/cm3. a) Find the volume of the kilogram standard of gold in cubic meter. b) In order to minimize the effect of surface contamination, the height of this golden standard kilogram is equal to its diameter. Find the height of this golden standard kilogram in mm.
Answers
Explanation:
a) Using the provided information about the density of gold, the sample size, thickness, and the following equations and comersion factors, find the area of the gold leaf:
\(V=l \cdot w \cdot h=A \cdot h\\m=\rho \cdot V\)
Gold \(_{\rho}=19.32 \mathrm{g} / \mathrm{cm}^{3}\)
\(1 \mu=10^{-6} \mathrm{m}\)
First, find the volume of the sample and then find the area of the sample.
\(V=\frac{m}{\rho}=\frac{27.6 \mathrm{g}}{19.32 \mathrm{g} / \mathrm{cm}^{3}} \cdot\left(\frac{0.01 \mathrm{m}}{1 \mathrm{cm}}\right)^{3}\) \(=\frac{1.429 \times 10^{-6} \mathrm{m}^{3}}\)
\(V=A \cdot h \rightarrow A=\frac{V}{h}=\frac{1.429 \times 10^{-6} \mathrm{m}^{3}}{10^{-6} \mathrm{m}} \approx 1.429 \mathrm{m}^{2}\)
b. Using the provided information from part \(a\) ), the radius of the cylinder, and the following equation for the volume of a cylinder, find the length of the fiber :
\(V=\pi r^{2} h \rightarrow h=\frac{V}{\pi r^{2}}\)
\(h=\frac{1.429 \times 10^{-6} \mathrm{m}^{3}}{\pi \cdot\left(2.5 \times 10^{-6} \mathrm{m}\right)^{2}} \approx 72778 \mathrm{m}\)
pls answer both! i ran out of
questions! thank you!
Use the References to access important values if needed for this question. The equilibrium constant, Kp, for the following reaction is 1.80 x 10-2 at 698 K. 2HI(g) → H₂(g) + I₂ (g) If an equilib
Answers
The equilibrium concentration of HI is 1.56 x 10-5 M and the equilibrium concentration of H₂ and I₂ is 7.8 x 10-6 M.
Given: The equilibrium constant, Kp, for the following reaction is 1.80 x 10-2 at 698 K.2HI(g) → H₂(g) + I₂ (g)
When equilibrium is reached, the concentration of H₂ is found to be 2.80 x 10-3 M. Calculate the equilibrium concentration of HI and I2.
Solution: Equilibrium constant, Kp = 1.80 x 10-2 at 698 K Since the equation is 2HI(g) → H₂(g) + I₂ (g),therefore the expression for Kp is given as,
Kp = [H₂] [I₂] / [HI]²
At equilibrium,[H₂] = 2.80 x 10-3 M We are to find the equilibrium concentration of HI and I2. Let the equilibrium concentration of HI be x and the equilibrium concentration of I2 be y. Molar concentration of H₂ = 2.80 x 10-3 M Using the equilibrium constant expression, Kp = [H₂] [I₂] / [HI]²= (2.80 x 10-3) (y) / (x)²= 2.80 x 10-3 (y) / (x²)---------------------eqn1We also know that,2HI(g) → H₂(g) + I₂ (g)Initially (before the reaction begins), concentration of HI = x and concentration of H₂ and I₂ are zero. Thus, initially, H₂ = 0and I₂ = 0At equilibrium, 2HI(g) → H₂(g) + I₂ (g).
Thus, initially the concentration of HI = x-moles. Then, for every 2 moles of HI that is converted, one mole of H₂ and one mole of I₂ are produced. So, the concentration of H₂ and I₂ at equilibrium would be x/2 moles. Because, for every 2 moles of HI that is converted, one mole of H₂ and one mole of I₂ are produced.[HI] = x M[H₂] = [I₂] = x/2 M Substituting the values in the expression derived above in eqn1,Kp = 1.80 x 10-2 = 2.80 x 10-3 (y) / (x²)= 2.80 x 10-3 (y) / x²x² = (2.80 x 10-3 y) / (1.80 x 10-2)= 0.15555y / 1Substituting the value of x² in the equation 1,1.80 x 10-2 = 2.80 x 10-3 (y) / 0.15555y1.80 x 10-2 = 18.00 y / 15555y1.80 x 10-2 = y / 865.3y = 1.56 x 10-5 M[H₂] = [I₂] = x/2 = (1.56 x 10-5 M) / 2= 7.8 x 10-6 M
∴ The equilibrium concentration of HI is 1.56 x 10-5 M and the equilibrium concentration of H₂ and I₂ is 7.8 x 10-6 M.
To know more about concentration visit
https://brainly.com/question/16551813
#SPJ11
Which of these best describes a scientific model?
A) the most recent version of a theory
B) a detailed description of a natural event
C) a way of explaining a complex concept D) a small version of a large object
Answers
What Group is this element in?
Please answer (^∇^)

Answers
Conclusion: Write a conclusion statement that addresses the following questions: How did your experimental absolute zero value compare to the accepted value? Does your data support or fail to support your hypothesis (include examples)? Discuss any possible sources of error that could have impacted the results of this lab. How do you think the investigation can be explored further? Post-Lab Reflection Questions Answer the reflection questions using what you have learned from the lesson and your experimental data. It will be helpful to refer to your chemistry journal notes. Answer questions in complete sentences. Why was the line of best fit method used to determine the experimental value of absolute zero? Which gas law is this experiment investigating? How does your graph represent the gas law under investigation? Using your knowledge of the kinetic molecular theory of gases, describe the relationship between volume and temperature of an ideal gas. Explain how this is reflected in your lab data. Pressure and number of moles remained constant during this experiment. If you wanted to test one of these variables in a future experiment, how would you use your knowledge of gas laws to set up the investigation?
Answers
Answer:
Brainliest pls
Explanation:
Although the information for the trial was not given, we can offer a summed-up reply in that while playing out an analysis to accomplish outright zero temperatures, the worth won't ever match the specific worth.
What is absolute zero?
Absolute zero is the lower furthest reaches of temperature. Considered the coldest conceivable temperature that can exist. Nonetheless, any endeavor to arrive at this temperature in a controlled climate has fizzled, researchers don't think reproducing this temperature is conceivable.
In this way, we can affirm that the worth of indisputably the zero tests didn't match the acknowledged worth. Assuming the speculation was that it would be troublesome or difficult to accomplish, then the information would uphold the theory, if not, it would neglect to do as such.
In outline, outright zero is a temperature that can't be reproduced in a lab, so the worth in this analysis doesn't match the acknowledged worth and there could be no further investigation to be done on this.
Question 5
Are the two triangles similar? If yes, then complete the similarity statement.
Select all that apply.
ASTRA
S
3
R 5 T
N
4
P
by
6
similarity
Q

Answers
It should be noted that the two triangles are Similar by AA Similarity Criteria.
What is Similarity?If the ratio of one triangle's two sides to another triangle's two sides is the same and both triangles' two sides inscribe the same angle, then two triangles are said to be similar.
AA Similarity Postulate occurs if two angles in one triangle are congruent to two angles in another triangle, then the two triangles are similar.
Learn more about Triangle on:
https://brainly.com/question/22470980
#SPJ1
identify the solid product that forms when the following aqueous solutions are mixed: cocl2(aq) na2s(aq) →
Answers
The molecule that results from combining the aqueous solutions as specified in the question is called CoS.
Give an example of a chemical equation.
The reactant entities are presented on the left hand side of a chemical equation, while the product entities are given on the right hand side. A chemical equation is a symbolic representation of a chemical reaction in the form of symbols and equations.
A chemical Equation Class 10 is what?
Chemical Formula: Chemical equations are used to represent chemical reactions using the symbols and substance formulae. For instance, A + B = C + D. A and B are referred to in this equation as the reactants, and C and D are referred to as the products.
To know more about aqueous solutions visit:
brainly.com/question/26856926
#SPJ4
in a hydrogen atom which transition would emit the longest wavelength light? select all that apply 1. n
Answers
The wavelength will be longer the higher the magnitude of the fundamental quantum number to which the electron is hopping. Thus, the longest wavelength is produced by the n = 6 to p = 8 transition.
How would one describe wavelengths?The length of a wave is expressed by its wavelength. The wavelength is the distance between one wave's crest and the following wave's crest. The wavelength can also be determined by measuring from the "trough" (bottom) of one wave to the "trough" of the following wave.
Why is wavelength significant and what is it?The distance that a wave repeats is measured by its wavelength. The letter lambda () stands for these recurrences, also known as wavelengths. Energy travels through a medium as waves (like the atmosphere, the vacuum, or even the ocean).
To know more about wavelength visit-
https://brainly.com/question/12924624
#SPJ4
If the pH of 1 liter of a 1.0 M carbonate buffer is 7.0, what is actual number of moles of H2CO3 and HCO3-? (pK = 6.37) moles of HCO3 - moles of H2CO3 0.86 I. II. 0.81 0.14 0.19 0.24 III. 0.76 IV. 0.19 0.81 V. 0.14 0.86 IV III V I
Previous question
Answers
The actual number of moles of H₂CO₃ is 0.2 moles and the actual number of moles of HCO₃⁻ is 0.8 moles. The correct answer is:
I. moles of HCO₃⁻ = 0.86 ;moles of H₂CO₃= 0.14
To solve this problem, we need to consider the equilibrium between H₂CO₃(carbonic acid) and HCO₃⁻ (bicarbonate ion) in a carbonate buffer system.
The Henderson-Hasselbalch equation is used to calculate the pH of a buffer system:
pH = pKa + log([A⁻]/[HA])
Here, [A⁻] represents the concentration of the conjugate base (HCO₃⁻ ) and [HA] represents the concentration of the acid (H₂CO₃).
Given that the pH of the carbonate buffer is 7.0, we can use the Henderson-Hasselbalch equation to determine the ratio of [A⁻] to [HA]. Let's calculate:
7.0 = 6.37 + log([HCO₃⁻ ]/[H₂CO₃])
Subtracting 6.37 from both sides:
7.0 - 6.37 = log([HCO₃⁻ ]/[H₂CO₃])
0.63 = log([HCO₃⁻ ]/[H₂CO₃])
Now we need to convert the logarithmic equation into an exponential form:
[HCO₃⁻ ]/[H₂CO₃] = \(10^{0.63\)
[HCO₃⁻ ]/[H₂CO₃] = 4.00
This means that for every 1 molecule of H₂CO₃, there are 4 molecules of HCO₃⁻ in the buffer solution.
Now, let's determine the number of moles of H₂CO₃ and HCO₃⁻ in the given 1-liter solution.
Assuming that the volume of the solution remains constant after dissociation:
[H₂CO₃] + [HCO₃⁻ ] = 1.0 M
We can substitute [HCO₃⁻ ] = 4[H₂CO₃] into the equation:
[H₂CO₃] + 4[H₂CO₃] = 1.0 M
5[H₂CO₃] = 1.0 M
[H₂CO₃] = 1.0 M / 5 = 0.2 M
Thus, the concentration of H₂CO₃is 0.2 M.
Since we have 1 liter of solution, the number of moles of H₂CO₃ is:
moles of H₂CO₃= concentration of H₂CO₃× volume of solution
= 0.2 M × 1 L
= 0.2 moles
As we calculated earlier, the ratio of [HCO₃⁻ ] to [H₂CO₃] is 4:1. Therefore, the number of moles of HCO₃⁻ is:
moles of HCO₃⁻ = 4 × moles of H₂CO₃
= 4 × 0.2 moles
= 0.8 moles
Therefore, the actual number of moles of H₂CO₃ is 0.2 moles and the actual number of moles of HCO₃⁻ is 0.8 moles.
Comparing these values to the given options, we find that the correct answer is:
I. moles of HCO₃⁻ = 0.86; moles of H₂CO₃= 0.14
Learn more about Carbonic Acid at
brainly.com/question/31314818
#SPJ4
Complete Question:
Find the attached image for complete Question.
