what pressure would a gas mixture in a 10.0 l tank exert if it were composed of 48.5 g he and 94.6 g co 2 at 398 k?
A. 7.02 atm
B. 39.6 atm
C. 58.7 atm
D. 32.6 atm
E. 46.6 atm
Answers
The pressure exerted by the gas mixture in the 10.0 L tank is approximately 46.6 atm (Option E).
To find the pressure exerted by a gas mixture in a 10.0 L tank, we can use the Ideal Gas Law: PV = nRT,
where P is pressure, V is volume, n is the number of moles, R is the Ideal Gas Constant (0.0821 L atm/mol K), and T is temperature in Kelvin.
Step 1: Calculate the number of moles (n) for each gas.
He: n = mass / molar mass = 48.5 g / (4.00 g/mol) = 12.125 mol
CO₂: n = mass / molar mass = 94.6 g / (44.01 g/mol) = 2.149 mol
Step 2: Add the moles of each gas to find the total number of moles (n_total) in the mixture.
n_total = 12.125 mol + 2.149 mol = 14.274 mol
Step 3: Plug the values into the Ideal Gas Law equation.
P = nRT / V = (14.274 mol)(0.0821 L atm/mol K)(398 K) / (10.0 L)
Step 4: Calculate the pressure.
P ≈ 46.6 atm
Therefore, the pressure exerted by the gas mixture in the 10.0 L tank is approximately 46.6 atm .
Learn more about pressure of a gas mixture : https://brainly.com/question/23486705
#SPJ11
Related Questions
Which substance is composed of only one type of atom? - Need asap!
Water
Gold
Salt
Sugar
Answers
Answer:
Gold
Hope it helps
have a good day
Increasing temperature can

Answers
When you add wind power to the electric power grid, which pollution issue are you most likely to help address in the U.S.? (Think about where energy comes from and the downsides of each fuel source)O Sulfur oxide (SO and SO2) emissions from coal-fired power plantsO energy technology O the crude oil
Answers
When you add wind power to the electric power grid, you are most likely to help address the issue of sulfur oxide (SO and SO2) emissions from coal-fired power plants in the U.S.
Coal-fired power plants are a significant source of electricity in the United States, but they also produce a large amount of air pollution, including sulfur oxide emissions. When sulfur oxides are released into the air, they can react with other chemicals to form acid rain, which can have harmful effects on the environment and human health.
By adding wind power to the electric grid, we can reduce the amount of electricity that is generated by burning coal, which can help to reduce sulfur oxide emissions from power plants. Wind power is a clean, renewable source of energy that does not produce any harmful air pollutants, making it a more environmentally friendly alternative to coal-fired power.
Overall, wind power can help to address the issue of sulfur oxide emissions from coal-fired power plants and can help to promote a cleaner and more sustainable energy future in the United States.
For more similar questions on wind power
brainly.com/question/25818989
#SPJ11
Can someone help me on this

Answers
I did this to the best of my ability. I have a hard time comprehending things sometimes so I’m so so so sorry if it’s wrong
What is the concentration of H+ at pH 4 in moldm-3 ?
Answers
Answer:
The pH of a solution is a measure of its concentration of hydrogen ions. The higher the concentration of H + ions in an acidic solution, the lower the pH. A pH of 1 represents a hydrogen ion concentration of 0.1 mol/dm 3.
...
pH and hydrogen ion concentration.
Concentration pH
0.001 mol/dm 3 3
0.0001 mol/dm 3 4
Explanation:
Answer:
0.0001
That the answer your welcom
use the law of conservation of mass to answer the questions. consider a hypothetical reaction in which a and b are reactants and c and d are products. if 25 g of a completely reacts with 26 g of b to produce 17 g of c, how many grams of d will be produced?
Answers
we can use this principle law of conservation of mass: 25 g + 26 g = 17 g + x g, where x represents the mass of d produced. Solving for x, we find that x = 34 g, so 34 g of d are produced.
The law of conservation of mass states that mass is conserved in a chemical reaction, meaning that the total mass of reactants must equal the total mass of products.
In a chemical reaction, the law of conservation of mass states that the total mass of reactants must equal the total mass of products. Using this principle, we can solve for the unknown mass of a product in a reaction. In this hypothetical reaction, 25 g of a and 26 g of b react to produce 17 g of c and some amount of d. We can write an equation to represent the relationship between the masses of reactants and products: 25 g + 26 g = 17 g + x g, where x represents the mass of d produced. To find the value of x, we can solve the equation: 25 g + 26 g = 17 g + x g, so x = 25 g + 26 g - 17 g = 34 g. Therefore, 34 g of d are produced in this reaction.
Learn more about law of conservation of mass here:
https://brainly.com/question/28711001
#SPJ4
The table shows the relative atomic masses of some common elements. Use this information to work out the relative formula mass of magnesium sulfate. The formula for magnesium sulfate is MgSO4.
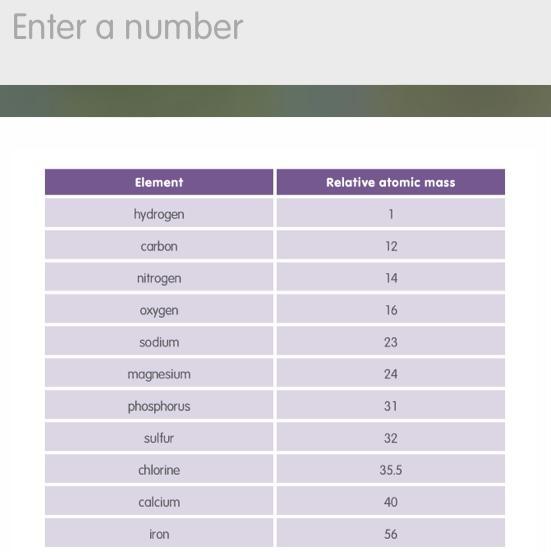
Answers
The relative atomic mass of Mg is 24.3, the relative atomic mass of S is 32.1, and the relative atomic mass of O is 16.0.
What is relative atomic mass?The relative atomic mass is a unit of measurement used in chemistry to express the mass of an atom of an element.It is defined as the ratio of the mass of an atom of an element to one twelfth of the mass of an atom of carbon-12.It is often used to compare the masses of atoms of different elements.The relative atomic mass is usually represented by the symbol "A" or "Ar" and is measured in atomic mass units (amu).It is important to note that the relative atomic mass is different from the atomic mass, which is the actual mass of an atom in grams.To calculate the relative formula mass of magnesium sulfate, we add up the relative atomic masses of each element in the formula:Mg = 24.3S = 32.14 x O = 4 x 16 = 64.0So, the relative formula mass of magnesium sulfate is 24.3 + 32.1 + 64.0 = 120.4.To learn more about relative atomic mass refer:
brainly.com/question/28882057
#SPJ1
Which high-energy bond is associated with the succinyl-CoA synthetase reaction?
A) acyl phosphate
B) thioester
C) phosphohistidine
D) mixed anhydride
E) All of the answers are correct
Answers
The high-energy bond associated with the succinyl-CoA synthetase reaction is A. acyl phosphate bond
Succinyl-CoA synthetase is an enzyme that catalyzes the conversion of succinyl-CoA to succinate, with the simultaneous synthesis of ATP or GTP from ADP or GDP, respectively. This reaction is an important step in the citric acid cycle, which is also known as the Krebs cycle or the tricarboxylic acid cycle.
The acyl phosphate bond in succinyl-CoA is a high-energy bond due to the resonance stabilization of the phosphate group, making it a favorable source of energy. When succinyl-CoA synthetase cleaves this bond, the energy released is used to phosphorylate the nucleoside diphosphate (ADP or GDP), forming a high-energy nucleoside triphosphate (ATP or GTP). Although options B, C, and D represent other types of high-energy bonds, they are not directly associated with the succinyl-CoA synthetase reaction. Therefore, the correct answer is A) acyl phosphate. So therefore the correct answer is A. Acyl phosphate bond, the high-energy bond associated with the succinyl-CoA synthetase reaction.
Learn more about enzyme at
https://brainly.com/question/30600790
#SPJ11
What is the percent by volume of isopropyl alcohol in a solution made by mixing 120 mL of the alcohol with enough water to make 350 mL of solution?
Answers
Answer: The percent by volume of isopropyl alcohol in a solution made by mixing 120 mL of the alcohol with enough water to make 350 mL of solution is 25.5%.
Explanation:
Given: Volume of solute = 120 mL
Volume of solvent = 350 mL
Now, total volume of the solution is as follows.
\(V_{total} = V_{solute} + V_{solvent}\\= 120 mL + 350 mL\\= 470 mL\)
Let us assume that 100 mL of solution is taken and the amount of isopropyl alcohol present in it is as follows.
\(\frac{V_{solute}}{V_{total}} \times 100 mL\\\frac{120 mL}{470} \times 100 mL\\= 25.53 mL\)
Hence, there is 25.53 mL isopropyl alcohol is present in 100 mL of solution. Therefore, %v/v is calculated as follows.
\(Percent (v/v) = \frac{25.53 mL}{100 mL}\\= 25.5%\)
Thus, we can conclude that the percent by volume of isopropyl alcohol in a solution made by mixing 120 mL of the alcohol with enough water to make 350 mL of solution is 25.5%.
Which statement explains how this type of fossil differs from a trace fossil?.
Answers
A body fossil preserves the physical remains of an organism, while a trace fossil preserves indirect evidence of the organism's activities or behavior. Both types of fossils contribute valuable information to our understanding of past life on Earth.
A fossil represents the preserved remains or traces of ancient organisms. The key difference between a body fossil and a trace fossil lies in the nature of the preserved evidence.
Body Fossil: A body fossil refers to the preserved remains of an organism's body parts, such as bones, teeth, shells, or plant structures. These fossils provide direct evidence of the organism's physical characteristics and are typically formed through processes like mineralization or petrification.
Trace Fossil: A trace fossil, also known as an ichnofossil, represents indirect evidence of ancient organisms' activities rather than their physical remains. Trace fossils include tracks, burrows, footprints, nests, and other trace marks left behind by organisms. These fossils provide insights into the behavior, movement, and ecological interactions of organisms rather than their anatomy.
for more questions on fossils
https://brainly.com/question/28122522
#SPJ8
What foods have peroxides?
Answers
Answer: Wheat flour, edible oil, egg white, lettuce preservative, meat or poultry marinade, vegetable antibacterial wash.
Explanation:
hydrogen peroxide is used as a bleaching agent in some food
I hope this helped
Identify the Lewis acid and Lewis base in each of the reactions. - C1- + AICI3 --> AICI4- ____ _____- BF3 +F- --> BF4-____ _____- NH3 + H+ --> NH4+____ _____
Answers
Each reaction's Lewis acid and Lewis base are as follows:
AlCl3 is the Lewis acid in the reaction, whereas Cl- is the Lewis base. F- is the Lewis base and BF3 is the Lewis acid in the reaction Cl- + AlCl3 --> AlCl4- BF3 + F- --> BF4-
The Lewis base in this reaction is NH3, and the Lewis acid is H+. NH3 + H+ --> NH4+
Explanation: A Lewis acid acts as an electron pair acceptor in a Lewis acid-base reaction, whereas a Lewis base acts as an electron pair donor. In the initial reaction, Cl- provides AlCl3 with a pair of electrons, which AlCl3 accepts to produce AlCl4-. As a result, AlCl3 is the Lewis acid and Cl- is the Lewis base. In the subsequent response, F- provides two pairs. BF3 takes the electrons and transforms them into BF4-. Hence, the Lewis bases are F- and BF3, respectively. In the third reaction, H+ absorbs a pair of electrons from NH3 and forms NH4+ as a result. As a result, the Lewis bases are NH3 and H+.
learn more about Lewis acid here:
https://brainly.com/question/15570523
#SPJ4
I have made a thermometer which measures temperature by the compressing and expanding of gas in a piston. I have measured that at 1000 C the volume of the piston is 30 L. What is the temperature outside if the piston has a volume of 5 L? What would be appropriate clothing for the weather? I
this is about boyle’s and charles law :)))
Answers
Answer:
The temperature is 298.5 K, which corresponds to 0.50°C.
A jacket would be appropriate clothing for this weather.
Explanation: Hope this helps:)
According to Charle's law, temperature outside the piston of a volume of 5 liters is -60.99 °C or 212.16 K. Appropriate clothing for the weather is woollen clothes.
What is Charle's law?Charles law is an experimental gas law which provides description about the behavior of gases which tend to expand on heating.It states that, at constant pressure, the temperature is in direct proportion with the volume that is,V∝T or V₁/T₁=V₂/T₂.The relation between volume and temperature is linear as the temperature increases volume also increases.
The graph of volume versus temperature is a straight line passing through the origin.
On substitution of values in the above formula,30/1273=5/T₂
∴T₂=1273×5/30=212.16 K or -60.99°C.
Thus ,the temperature of the gas at 5 liter volume is -60.99°C.
Learn more about Charle's law,here:
https://brainly.com/question/16927784
#SPJ2
Select the correct choice there's only one answer.

Answers
Answer:
Option C is the correct option.The Valence electrons in S
Explanation:
Sulphur has highest effective nuclear charge as in a period there is increase in effective nuclear charge from left to right.Sulphur is in the end in a period leaving Mg and Al in left side.
Hope this helps....
Good luck on your assignment...
Uneven heat distribution
Answers
How does electron pair repulsion determine the molecular shape/molecule geometry?.
Answers
The number of valence electron pairs in the outermost shell, as determined by the valence shell electron repulsion theory (VSEPR), determines the molecular shape.
By analyzing the repulsion between bond electron pairs in the outermost electron shell, a process known as the molecular shape can determine the shape of a molecule. Because most physical and chemical properties are influenced by molecular shape, it is crucial to study molecular shape or geometry.
The foundation of VSEPR is minimizing the strength of the electron-pair repulsion surrounding the central atom under consideration. The foundation of the VSEPR theory is the notion that the geometry (shape) of a molecule is primarily determined by the repulsion between the pairs of electrons surrounding a central atom.
Learn to know more about Electron pair repulsion on
https://brainly.com/question/10271048
#SPJ4
What is the
calibration of this
Erlenmyer flask?
calibration =
A. 10 mL
B. 125 mL
C. 50 mL
D. 25 mL
difference between 2 marked values
# of spaces between marked values
125 ml
-100
75
50

Answers
The 25 mL is the calibration of this Erlenmeyer flask. so, option (b) is correct.
What is calibration ?
The process of calibrating a measurement instrument involves comparing it to a traceable reference device and documenting the comparison. A "calibrator" is another name for the reference standard. It stands to reason that the reference is more accurate than the calibration instrument.
What is Erlenmeyer flask?
Erlenmeyer flasks are used to mix, heat, cool, incubate, filter, store, and perform various liquid-handling operations on liquids. For titrations and boiling liquids, their sloping sides and narrow necks enable the contents to be blended by swirling without the risk of spillage.
Therefore, 25 mL is the calibration of this Erlenmeyer flask.
Learn more about calibration from the given link.
https://brainly.com/question/525672
#SPJ1
How many 4d electrons would be predicted in the ground state for the following elements?a. zirconiumb. cadmiumc. iridiumd. iron
Answers
In order to answer the question first we must write the atomic number of each element:
Zirconium (Zr): 40
Cadmium (Cd): 48
Iridium (Ir): 77
Iron (Fe): 26
Then, we have to complete the distribution of electrons in each orbital for each atom:
The first 4 levels have the following distribution:
Level1: 1s
Number of electrones: 2
Level 2: 2s, 2p
Number of electrones 8 (2 in the s orbital and 6 in the p orbitals).
Level3: 3s, 3p, 3d
Number of electrones 18 (2 in the s orbital, 6 in the p orbital and 10 in the d orbitals)
Level 4: 4s, 4p, 4d, 4f
Number of electrones 32 (2 in the s orbital, 6 in the p orbitals, 10 in the d orbitals and 14 in the f orbitals)
The order in which the orbitlas are completed depends on the energy of each level. For example the 4s orbitals will be completed before the 3d orbitals because their energy is lower.
The order is as follows:
1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p, 5s, 4d, 5p...
Now, knowing the atomic number we can answer the question:
For Zirconium (total 40 electrones):
\(1s^2,2s^2,2p^6,3s^2,3p^6,4s^2,3d^{10},4p^6,5s^2,4d^2\)2 electrones are predicted in the 4d orbital
For Cadmium (total 48 electrones):
\(1s^2,2s^2,2p^6,3s^2,3p^6,4s^2,3d^{10},4p^6,5s^2,4d^{10}^{}\)10 electrones are predicted in the 4d orbital
For iridium, as it has an atomic number higher than Cadmium we can predict tha it also complets the 4d orbital, then it has also 10 electrones in it.
For iron (total 26 electrones)
\(1s^2,2s^2,2p^6,3s^2,3p^64s^2,3d^6\)Iron has no electrones in the 4d orbitals
Earth's gravitational potential energy: GPE = mgh = Gravity (9.81m/s2) *
Mass (kg) x Height (m)
Kinetic energy: KE= Imov?
How do the mass and speed of an object affect the kinetic energy?
Answers
Answer:
as mass and the square of speed is directly proportional to the the kinetic enegry so, more there will be mass and speed there will be more kinetic enegry.
The diagram illustrates an experiment where baking soda was
added to a container of vinegar. After five minutes, the balloon on
the top of the bottle started expanding. Explain what happened and
how this experiment demonstrates the Law of Conservation of
Matter.
Answers
A reaction between an acid and a base occurs when vinegar and baking soda are combined. Baking soda serves as the base, and vinegar is the acid. A gas known as carbon dioxide bubbles and foams as a result of this reaction between the two. Because there is nowhere else for this gas, it expands the balloon, resulting in the self-inflating balloon.
What happens when vinegar and baking soda are combined in a bottle and a balloon is inserted into the bottle's mouth?SCIENCE: Carbon dioxide is a gas produced when baking soda and vinegar are combined. The gas starts to extend in the jug and begins to expand the inflatable. The balloon will expand in size the more gas produced.
What is the balloon experiment's conclusion?Because their particles travel at high speeds in all directions, gases expand quickly. Since the carbon dioxide gas has nowhere else to go, it begins to fill the balloon as it fills the bottle. The balloon expands as the carbon dioxide gas fills it.
To learn more about carbon dioxide here
https://brainly.com/question/3049557
#SPJ1
a sample of sulfur has a mass of 28.5 grams. What is this samples volume in ml?
Answers
Answer:
Incorrect in order to find Volume there must be a Density
LAB REPORT
Lab: Charles law
I don’t know how to do this someone pls help I’m so behind I’m crying

Answers
Answer:
The physical principle known as Charles' law states that the volume of a gas equals a constant value multiplied by its temperature as measured on the Kelvin scale (zero Kelvin corresponds to -273.15 degrees Celsius
Explanation:
What distinguishes an element from all other elements
Answers
Answer:
Elements are distinguished from one another by their atomic numbers. No two elements have the same atomic number. It is the atomic number, and therefore the number of protons in its atomic nuclei, that define an element.
!
Hormones keep our bodies in a state of
balance known as
(a)metabolism
(b)homeostasis
Answers
PLEASE URGENT HELP!! 30 POINTS!!!!!!

Answers
Answer:
CH³–C=CH——CH³–CH=CH²
Explanation:
THIS IS PROPYNE TO PROPENE
10. what is the magnitude of change in the freezing point of 1.00 l of water when one mole of nacl is added? when two moles of nacl are added?
Answers
The magnitude of change in the freezing point of 1.00 L of water when one mole of NaCl is added is 1.86°C.
The magnitude of change in the freezing point of 1.00 L of water when two moles of NaCl is added is 3.72°C.
When one mole of NaCl is added to 1.00 L of water, the magnitude of change in the freezing point is calculated as follows:
Change in freezing point = Kf × molality of solute
For water, the value of the freezing point depression constant (Kf) is 1.86°C/m. Since we are dealing with one mole of NaCl in 1.00 L of water, the molality of the solution can be calculated as follows:
Molality (m) = Number of moles of solute (n) / Mass of solvent (kg)
Since we have 1 mole of NaCl in 1.00 L of water, the mass of water is 1.00 kg. Therefore, the molality is:
Molality (m) = 1 mole / 1.00 kg = 1 mol/kg
Now we can calculate the change in freezing point:
Change in freezing point = Kf × molality of solute = 1.86°C/m × 1 mol/kg = 1.86°C
Different calculations will be performed when two moles of NaCl are added. The calculation will be similar to the above mentioned except the number of moles of NaCl will change from 1 to 2. The molality of the solution can be calculated as follows:
Molality (m) = Number of moles of solute (n) / Mass of solvent (kg)
Molality (m) = 2 mole / 1.00 kg = 2 mol/kg
Therefore, the change in freezing point will be:
Change in freezing point = Kf × molality of solute = 1.86°C/m × 2 mol/kg = 3.72°C
Learn more about freezing point here: https://brainly.com/question/30121086
#SPJ11
Sucrose has the molecular formula c12h22o11. if a sucrose sample contains 9.0x10^24 atoms of hydrogen, how many molecules of sucrose are present in the sample?
Answers
Sucrose has the molecular C₁₂H₂₂O₁₁ . if a sucrose sample contains 9.0x10²⁴ atoms of hydrogen, There are 1.4370 x10²⁶ molecules of sucrose are present in the sample.
Sucrose C₁₂H₂₂O₁₁ contain C₁₂H₂₂O₁₁ Hydrogen
First we should how how many percentage of hydrogen in sucrose
The following formula will be used to determine the percentage of elements present in a molecule:
(Mass of Element / Mass of Molecule) / 100 equals the percentage of an element.
Sucrose's molecular weight is.
= (12)12 + (1)22 + (16)11,
where 12 is the mass of an atom of carbon.
Hydrogen's atomic mass is 1, thus.
16 = Oxygen Atomic Mass
mass sucrose = 144 + 22 + 176
mass sucrose = 342 g/mol
% of H = (22 / 342) ×100 = 6.43 %
to calculate how many molecules sucrose are present we can compare percentage of hydrogen to % sucrose
% hydrogen = % sucrose
atom hydrogen molecules of sucrose
6.43 % = 100%
9.0x10²⁴ molecules of sucrose
molecules of sucrose = 1.4370 x10²⁶ molecules
Therefore, There are 1.4370 x10²⁶ molecules of sucrose are present in the sample.
Learn more about number of molecule at https://brainly.com/question/28027501
#SPJ4
what is the function of acetylcholinesterase? where is it found?
Answers
Acetylcholinesterase is an enzyme that breaks down the neurotransmitter acetylcholine into choline and acetate. This process is called hydrolysis and terminates the action of acetylcholine in the nervous system.
Acetylcholinesterase is found in the synaptic cleft, which is the narrow gap between nerve cells where neurotransmitters are released and received. It is also found in other tissues, including the neuromuscular junction, red blood cells, and the central nervous system. In the neuromuscular junction, acetylcholinesterase plays a critical role in terminating the action of acetylcholine, which is necessary for proper muscle function. The proper functioning of acetylcholinesterase is important for normal nervous system function and various biological processes, including cognition, memory, muscle movement, and other bodily functions. Dysfunction of acetylcholinesterase has been linked to various medical conditions, including Alzheimer's disease, myasthenia gravis, and organophosphate poisoning.
Learn more about Acetylcholinesterase here:
https://brainly.com/question/9240016
#SPJ4
isotopes with an even number of both protons and neutrons are generally stable. true or false
Answers
The statement "Isotopes with an even number of both protons and neutrons are generally stable" is TRUE.
Isotopes with an even number of protons and neutrons are known as isotopes. A single element can have a different number of neutrons; such elements are known as isotopes. A nucleus containing a different number of neutrons will have a different mass number than the element's standard atomic number since the mass number is equal to the number of neutrons plus the number of protons in the nucleus.
There are 275 isotopes recognized for 81 stable elements, while there are 50 isotopes for elements that are radioactive and occur naturally.
Isotopes with an even number of both protons and neutrons tend to be more stable, as the strong nuclear force and the electromagnetic force in a nucleus cancel out, causing a more stable nucleus. The vast majority of stable elements have a nearly equal number of neutrons and protons, though there are some exceptions, such as beryllium-8 and helium-3.
Learn more about the isotopes from the given link-
https://brainly.com/question/13604636
#SPJ11
When a spoonful of salt is dissolved in a pan filled with water, the water is called the...
Select one:
a. solute.
b. solution.
c. solvate.
d. solvent.
Answers
Answer:
d
Explanation:
it is called the solvent. let me know if this is wrong.
Answer:
d
Explanation:
the solvent is the liquid that the salt is dissolved in(the water) the solute is what is dissolved in the solvent(the salt) the solution is the outcome of the experiment.