Answers
Answer:
Mitochondria is the powerhouse of the cell, but don't let this distract you from the fact that Mr. Krabs sold SpongeBob's soul for 62 cents
Answer:
mitochondria
Explanation:
Related Questions
Determine the number of carbon atoms in 1.00 kg of carbon dioxide.
(Enter in scientific notation)
—— can someone pls show me the steps, I’m struggling :((
Answers
Answer:
1.37 × 10²⁵ atoms
Explanation:
Step 1: Given data
Mass of CO₂ (m): 1.00 kg
Step 2: Convert "m" to grams
We will use the relationship 1 kg = 10³ g.
1.00 kg × (10³ g/1 kg) = 1.00 × 10³ g
Step 3: Calculate the number of moles corresponding to 1.00 × 10³ g of CO₂
The molar mass of CO₂ is 44.01 g/mol.
1.00 × 10³ g × (1 mol/44.01 g) = 22.7 mol
Step 4: Calculate the moles of C in 22.7 moles of CO₂
The molar ratio of C to CO₂ is 1:1. The moles of C are 1/1 × 22.7 mol = 22.7 mol.
Step 5: Calculate the number of atoms of C in 22.7 moles of C
We will use Avogadro's number: there are 6.02 × 10²³ atoms of C in 1 mole of C.
22.7 mol × (6.02 × 10²³ atoms/1 mol) = 1.37 × 10²⁵ atoms
In a common reaction in living things, glucose combines with oxygen to form carbon dioxide and water vapor. In an experiment on this reaction, the masses of glucose, oxygen, and carbon dioxide are measured. This data table shows the measurements. What is the mass of the water vapor? please help
Answers
Multiply the number of moles of glucose by the molar mass of water (18.015 g/mol) to find the mass of water vapor produced.
To determine the mass of water vapor produced in the reaction, we need to analyze the data provided in the data table. The reaction mentioned is the process of cellular respiration, where glucose (C6H12O6) combines with oxygen (O2) to produce carbon dioxide (CO2) and water (H2O). We can use the principle of conservation of mass to calculate the mass of water vapor produced.The data table should provide the initial masses of glucose and oxygen, as well as the final mass of carbon dioxide. From these measurements, we can calculate the mass of water vapor.First, determine the number of moles of glucose and oxygen by dividing their respective masses by their molar masses. Then, use the balanced equation for cellular respiration to determine the mole ratios between glucose, oxygen, carbon dioxide, and water.
The balanced equation for cellular respiration is:
C6H12O6 + 6O2 -> 6CO2 + 6H2O
Based on this equation, we know that for every mole of glucose, 6 moles of water are produced.In conclusion, by using the principles of conservation of mass and stoichiometry, you can calculate the mass of water vapor produced in the reaction based on the given measurements.
for such more questions on moles
https://brainly.com/question/15356425
#SPJ8
Question 12 of 25
Which of the following is the best summary of Avogadro's law?
A. Equal amounts of all gases occupy 22.4 L at all conditions.
B. Equal amounts of all gases have the same volume at the same
conditions,
C. Every gas has a different volume at STP.
D. The volume a gas occupies depends on gas type.
SUBMIT
Answers
Answer:
B. Equal amounts of all gases have the same volume at the same
conditions
Explanation:
Amedo Avogadro found the relationship between volume of a gas and the number of molecules contained in the volume.
The law states that "equal volumes of all gases at the same temperature and pressure contains equal number of molecules or moles".
The law describes the behavior of gases when involve in chemical reactions. It enables one to change over at will in any statement about gases from volumes to molecules and vice versa.
So, the right option is B which implies that equal amounts of all gases have the same volume at the same conditions.
Part B
Next, you’ll test your hypothesis from part A by examining the reaction times of vinegar and baking soda in water at four different temperatures. You’ll carry out the reaction using water at room temperature (about 25°C), 40°C, 60°C, and 80°C. Make sure that you use the same amounts of vinegar and baking soda for all three three trials.
Gather all the materials, and perform these steps for each trial:
Heat at least
cup (60 milliliters) of water to the required temperature (refer to the data table). Water may be heated on a stove, on a hot plate, or in a microwave oven.
Measure and record the actual temperature of the water.
Measure 1 tablespoon (15 milliliters) of the water into the cup.
Add
teaspoon (1.5 grams) baking soda to the water, and stir until it is dissolved. The solution will be clear.
Measure 1 tablespoon (15 milliliters) of vinegar, but do not pour it into the cup yet.
Very quickly, do all of the following:
a. Pour the measured vinegar into the cup.
b. Start the stopwatch.
c. Stir or carefully swirl the substances in the cup.
The chemical reaction will produce bubbles. You’ll be able to see the bubbles and hear them pop. Watch and listen for when the reaction stops. When it looks and sounds like it has finished, stop the stopwatch.
Record the reaction time in the data table.
Discard the solution down the drain, and rinse the cup.
Repeat steps 1–9 of this procedure, doing three trials for each water temperature. Record the average temperature and reaction time for each set of the three trials. Read this math review to know how to calculate average of a data set.
Answers
The reaction time decreases as the temperature increases of the reaction mixture increases.
A sample record of results is:
Temperature (°C) Trial 1 Trial 2 Trial 3 Average25°C 11 seconds 11 seconds 11 seconds 11 seconds40°C 8 seconds 8 seconds 8 seconds 8 seconds60°C 5 seconds 5 seconds 5 seconds 5 seconds80°C 3 seconds 3 seconds 3 seconds 3 secondsWhat is the effect of an increase in temperature on reaction time?An increase in temperature leads to an increase in reaction rate or a decrease in reaction time.
The increase in temperature provides more thermal energy to the reactant molecules, which leads to an increase in the average kinetic energy of the molecules. As a result, more reactant molecules have sufficient energy to overcome the activation energy barrier and undergo successful collisions, leading to an increased reaction rate.
Learn more about reaction time at: https://brainly.com/question/26142029
#SPJ1
PLEASE HELP!!!
2C₂H6 (g) + 702 (g) —> 4CO2 (g) + 6H₂O(g)
If 10.0 liters of ethane gas are used, how many liters of oxygen gas will be
needed for the above reaction at STP?
A: 35.0 liters
B: 2.85 liters
C: 70.0 liters
D: 1.4 liters

Answers
The volume (in liters) of oxygen gas, O₂ that will be needed for the above reaction at STP is 35 liters (option A)
How do i determine the volume of oxygen gas needed?The volume of oxygen gas needed for the reaction can be obtain as shown below:
Balanced equation:
2C₂H₆(g) + 7O₂(g) —> 4CO₂(g) + 6H₂O(g)
From the question given above, we were told that the reaction occur at standard temperature and pressure, STP. Thus we can obtain the volume of oxygen needed as follow::
2 liters of C₂H₂ reacted with 7 liters of O₂
Therefore
10 liters of C₂H₂ will react = (10 liters × 7 liters) / 2 liters = 35 liters of O₂
Thus, we can conclude that the volume of oxygen gas needed for the reaction is 35 liters (option A)
Learn more about volume:
https://brainly.com/question/9614052
#SPJ1
Which element has the largest atomic radius?
4 be
56 ba
31 ga
9 f
Answers
Answer:
Ba
Explanation:
bc it = 268 pm
melting points of lipids are strongly influenced by the length and degree of unsaturation of hydrocarbon chain.justify this statement?
Answers
Answer
The properties of fatty acids and of lipids derived from them are markedly dependent on chain length and degree of saturation. Unsaturated fatty acids have lower melting points than saturated fatty acids of the same length. For example, the melting point of stearic acid is 69.6°C, whereas that of oleic acid (which contains one cis double bond) is 13.4°C. The melting points of polyunsaturated fatty acids of the C18 series are even lower. Chain length also affects the melting point, as illustrated by the fact that the melting temperature of palmitic acid (C16) is 6.5 degrees lower than that of stearic acid (C18). Thus, short chain length and unsaturation enhance the fluidity of fatty acids and of their derivatives.
A student made a graph plotting the progress of a reaction over time. The student forgot to label the y-axis of the graph.
A graph is shown with two graph lines. One graph line starts at a higher position on the y axis and slopes downwards towards the right. The other graph line starts at a lower position on the y axis and slopes upwards towards the right. The two graph lines stop short of intersecting each other and continue as separate lines which gradually become straight and parallel to the x axis. A vertical line is shown at a point where the two graph lines finally became parallel to the x axis. This vertical line is labeled equilibrium. The title on the x axis is Time and an arrow pointing towards the right is shown above Time. The title on the y axis is left blank.
What best explains the label that the student should use on the y-axis? (5 points)
Group of answer choices
Amount, because as the amount of product decreases, the amount of reactant increases over time.
Reaction rate, because forward and backward reaction become equal at equilibrium.
Amount, because the amounts of reactants and products become constant after equilibrium is reached.
Reaction rate, as the rate of forward reaction increases and rate of backward reaction decreases over time.

Answers
Answer:
C
Explanation:
Recall that, at equilibrium, the concentration of the reactants and products stop changing because both the forward and reverse reaction rates are equal.
Therefore, Choice C is correct.
Choice A is incorrect because there is no indication that the upper line describes the products and the lower line describes the reactants.
Choice B is incorrect because the statement, while true, is not reflected by the graph. (The two lines after equilibrium are not at the same y-value.)
Choice D is incorrect because the forward reaction rate generally decreases as time increases due to the decrease in reactant concentration(s).
PLEASE HELP
C. Balance these fossil-fuel combustion reactions. (1 point)

Answers
Answer:
C8H18(g) + 12.5O2(g) -> __8__CO2(g) + 9H2O(g) + heat
CH4(g) + _2___O2(g) -> ____CO2(g) + _2___H2O(g) + heat
C3H8(g) + _5___O2(g) -> _3___CO2(g) + __4__H2O(g) + heat
2C6H6(g) + __15__O2(g) -> __12__CO2(g) + __6__H2O(g) + heat
Explanation:
I hope it helps!
Fossil fuels are natural energy resources formed from the remains of ancient plants and organisms, such as coal, oil, and natural gas, which are burned to release energy.
Balanced reactions are as follows:
C₈H₁₈ (g) + 12.5O₂ (g) → 8CO₂ (g) + 9H₂O(g) + heat
CH₄ (g) + 2O₂ (g) → 1CO₂ (g) + 2H₂O (g) + heat
C₃H₈ (g) + 5O₂ (g) → 3CO₂ (g) + 4H₂O (g) + heat
C₆H₆ (g) + 7.5O₂ (g) → 6CO₂ (g) + 3H₂O (g) + heat
A combustion reaction, also known as a combustion process, is a chemical reaction in which a substance combines with oxygen to produce heat and light. It involves the rapid oxidation of a fuel in the presence of an oxidizer, typically atmospheric oxygen. The reaction releases energy in the form of heat and light.
The general equation for a combustion reaction can be represented as: fuel + oxygen → carbon dioxide + water + energy.
Combustion reactions are essential for various processes, including the burning of fossil fuels for energy production, combustion engines in vehicles, and even the process of respiration in living organisms.
Know more about combustion here:
https://brainly.com/question/34231206
#SPJ2
What is the length of the paper clip in cm
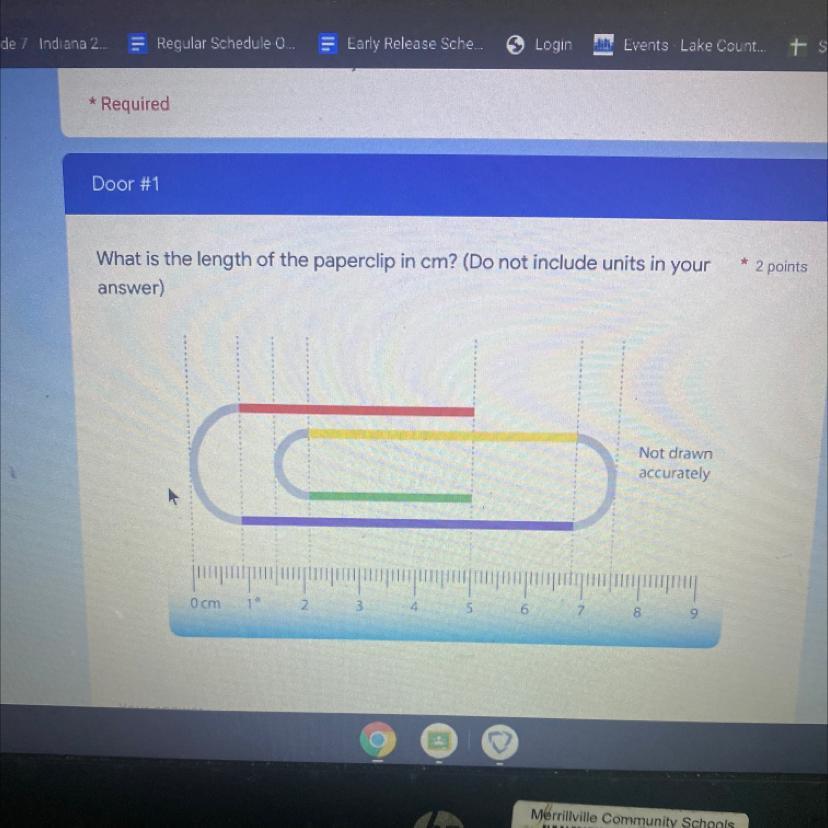
Answers
Answer:
24.5 cm
Explanation:
It would be really complicated to type out, so I've attached an image of how I solved this:
*I separated the paperclip into different sections, figured out the length of those sections, and added them together.
(sorry that my work isn't the neatest)

6) The density of ammonia gas (NHs) in a 6.0 L container at a pressure of 820 mm Hg and a g/L.
Answers
The density of ammonia gas in the 6.0 L container at a pressure of 820 mm Hg is approximately 0.805 g/L.
To determine the density of ammonia gas (NH3) in a 6.0 L container at a pressure of 820 mm Hg, we need to use the ideal gas law equation, which relates pressure, volume, number of moles, and temperature for a given gas.
The ideal gas law equation is:
PV = nRT
Where P is the pressure, V is the volume, n is the number of moles, R is the ideal gas constant, and T is the temperature in Kelvin.
Since we are given the pressure (820 mm Hg), volume (6.0 L), and assuming standard temperature and pressure (STP), we can use the values for R (0.0821 L·atm/(mol·K)) and convert the pressure to atm by dividing by 760 (1 atm = 760 mm Hg).
820 mm Hg / 760 mm Hg/atm = 1.08 atm
Now we can rearrange the ideal gas law equation to solve for density (d):
d = (P * M) / (RT)
Where M is the molar mass of ammonia (NH3), which is approximately 17.03 g/mol.
Substituting the values, we have:
d = (1.08 atm * 17.03 g/mol) / (0.0821 L·atm/(mol·K) * 298 K)
Simplifying the equation, we find:
d ≈ 0.805 g/L
Therefore, the density of ammonia gas in the 6.0 L container at a pressure of 820 mm Hg is approximately 0.805 g/L.
For more question on density
https://brainly.com/question/26364788
#SPJ8
What does this image represent?
a) alcohol group
b) carbonyl group
c) ether group
d) hydroxyl group

Answers
Explanation:
It represent Alcohol group (—OH)
Two asteroids are 75,000 m apart one has a mass of 8 x 10^7 N what is the mass of the other asteroid
Answers
The mass of the asteroid is C. 1.2 x \(10^{12}\) Kg
To find the mass of the other asteroid, we can rearrange the equation for the gravitational force between two objects:
F = (G * m1 * m2) / \(r^{2}\)
where F is the force of gravity, G is the gravitational constant, m1 and m2 are the masses of the two asteroids, and r is the distance between them.
Given that the distance between the asteroids is 75000 m, the force of gravity between them is 1.14 N, and one asteroid has a mass of 8 x \(10^{7}\) kg, we can substitute these values into the equation and solve for the mass of the other asteroid (m2):
1.14 N = (6.67430 × \(10^{-11}\) N \(m^{2}\)/\(Kg^{2}\) * 8 x \(10^{7}\) kg * \(m2\)) / \((75000 m)^{2}\)
Simplifying and solving the equation, we find that the mass of the other asteroid (m2) is approximately 1.2 x \(10^{12}\) kg. Therefore, Option C is correct.
The question was incomplete. find the full content below:
Two asteroids are 75000 m apart one has a mass of 8 x \(10^{7}\) kg if the force of gravity between them is 1.14 what is the mass of the asteroid
A. 3.4 x \(10^{11}\) kg
B. 8.3 x \(10^{12}\) kg
C. 1.2 x \(10^{12}\) kg
D. 1.2 x \(10^{10}\) kg
Know more about gravitational force here:
https://brainly.com/question/72250
#SPJ8
What is the shape of a molecule that has 3 atoms bonded to a central atom and no lone pairs of electrons?
Answers
Answer and Explanation: A molecule consisting of central atom with three bond pairs and one lone pair of electrons will have trigonal pyramidal shape.
7: How many times can a cell divide?
Answers
Explanation:
60 times and it matters why they need to in order to divide
share the imporatant lesson that you have learned in organic chem
Answers
We study the reactions that chemists utilise to create bizarre carbon-based structures in organic chemistry.
The study of the makeup, properties, and responses of organic compounds including organic materials, or matter in any of its many forms that contains carbon atoms, is the subject of the branch of science known as organic chemistry. Their structural formula is determined by study of structure.
We will study the reactions that chemists utilise to create bizarre carbon-based structures in organic chemistry, in addition to the analytical techniques used to characterise them. We'll also consider the molecular reaction mechanisms that are driving those reactions.
To know more about organic chemistry, here:
https://brainly.com/question/14623424
#SPJ1
If you want to melt ice into liquid water, does the ice absorbs heat or release heat? Is this endothermic or exothermic?
Answers
Answer:
Endothermic
Explanation:
This would be an endothermic reaction, due to the fact that the water would be signifigantly warmer then the ice cube, which causes it to melt.
Answer:
absorbs heat, which makes it endothermic.
Explanation:
Basically melting ice is it in windows or Mac reaction makes the ice absorbs heat energy which causes a change to occur
Starting with 0.3500 mol CO(g) and 0.05500 mol COCl2(g) in a 3.050 L flask at 668 K, how many moles of CI2(g) will be present at equilibrium?
CO(g) + Cl2(g)》COCl2(g)
Kc= 1.2 x 10^3 at 668 K
Answers
At equilibrium, the number of moles of \(Cl_2\) (g) will be 0.2025 mol.
1: Write the balanced chemical equation:
\(C_O\)(g) + \(Cl_2\)(g) ⟶ \(C_OCl_2\)(g)
2: Set up an ICE table to track the changes in moles of the substances involved in the reaction.
Initial:
\(C_O\)(g) = 0.3500 mol
\(Cl_2\)(g) = 0.05500 mol
\(C_OCl_2\)(g) = 0 mol
Change:
\(C_O\)(g) = -x
\(Cl_2\)(g) = -x
\(C_OCl_2\)(g) = +x
Equilibrium:
\(C_O\)(g) = 0.3500 - x mol
\(Cl_2\)(g) = 0.05500 - x mol
\(C_OCl_2\)(g) = x mol
3: Write the expression for the equilibrium constant (Kc) using the concentrations of the species involved:
Kc = [\(C_OCl_2\)(g)] / [\(C_O\)(g)] * [\(Cl_2\)(g)]
4: Substitute the given equilibrium constant (Kc) value into the expression:
1.2 x \(10^3\) = x / (0.3500 - x) * (0.05500 - x)
5: Solve the equation for x. Rearrange the equation to obtain a quadratic equation:
1.2 x \(10^3\) * (0.3500 - x) * (0.05500 - x) = x
6: Simplify and solve the quadratic equation. This can be done by multiplying out the terms, rearranging the equation to standard quadratic form, and then using the quadratic formula.
7: After solving the quadratic equation, you will find two possible values for x. However, since the number of moles cannot be negative, we discard the negative solution.
8: The positive value of x represents the number of moles of \(Cl_2\)(g) at equilibrium. Substitute the value of x into the expression for \(Cl_2\)(g):
\(Cl_2\)(g) = 0.05500 - x
9: Calculate the value of \(Cl_2\)(g) at equilibrium:
\(Cl_2\)(g) = 0.05500 - x
\(Cl_2\)(g) = 0.05500 - (positive value of x)
10: Calculate the final value of \(Cl_2\) (g) at equilibrium to get the answer.
Therefore, at equilibrium, the number of moles of \(Cl_2\) (g) will be 0.2025 mol.
For more such questions on equilibrium, click on:
https://brainly.com/question/517289
#SPJ8
Differences between voltage, current and resistance?
Answers
Answer:
Voltage is the measure of electric potential energy per unit charge, current is the flow of electric charge through a circuit, and resistance is the property of a material that opposes the flow of electric current.
Ohm's Law relates these three concepts by stating that current is directly proportional to voltage and inversely proportional to resistance.
Hope this helps!
what are thetypes of luminous flame
Answers
Types of luminous flames:
1. Yellow Luminous Flame
2. Smoky Luminous Flame
3. Orange Luminous Flame
4. Blue Luminous Flame
Luminous flames are characterized by their visible glow, which is caused by the incomplete combustion of fuel. The presence of soot particles in the flame causes the emission of light. There are different types of luminous flames, which can be classified based on their fuel composition and burning conditions. Here are some common types of luminous flames:
1. Yellow Luminous Flame: This is the most common type of luminous flame, often seen in open fires, candles, and gas stoves. It appears yellow due to the presence of soot particles in the flame. Yellow flames indicate incomplete combustion of hydrocarbon fuels, such as methane, propane, or natural gas. The high carbon content in these fuels leads to the formation of soot, which emits visible light.
2. Smoky Luminous Flame: This type of flame is characterized by a significant amount of black smoke and soot production. It is commonly observed in poorly adjusted or malfunctioning burners or engines. The excessive presence of unburned fuel in the flame results in incomplete combustion and the emission of dark smoke particles.
3. Orange Luminous Flame: An orange flame indicates a higher combustion temperature compared to a yellow flame. It is often seen in more efficient burners or when burning fuels with a higher carbon content, such as oil or diesel. The higher temperature helps in burning more of the carbon particles, reducing the amount of soot and making the flame appear less yellow.
4. Blue Luminous Flame: A blue flame is typically associated with complete combustion. It indicates efficient burning of fuel, resulting in minimal soot formation. Blue flames are commonly observed in gas burners or Bunsen burners. The blue color is a result of the combustion of gases, such as methane, in the presence of sufficient oxygen.
It's important to note that the luminosity of a flame can vary depending on factors such as fuel-air mixture, combustion temperature, and the presence of impurities. Achieving complete combustion and minimizing the production of soot is desirable for efficient and cleaner burning processes.
for more questions on luminous
https://brainly.com/question/27163038
#SPJ8
Would you rather wear
Socks in sandals or wear shoes without socks
Answers
Answer:
i always wear shoes without socks
Explanation:
Which type of rock does Florian predict these pieces will change into when subjected to heat and pressure?
Answers
Answer:
The Florian predict the metamorphic rocks because it changes when subjected to heat and pressure.
Explanation:
i hope this helps
Fructose-1-phosphate can be hydrolyzed into fructose + inorganic phosphate (Pi) with a ΔG° of –16.0 kJ/mol. If ATP can be hydrolyzed into ADP + Pi with a ΔG° of –30.5 kJ/mol, what is the free energy change for the reaction of fructose + ATP → fructose 1-phospate + ADP
Answers
ΔG° (overall reaction) = ΔG° (sum of products) - ΔG° (sum of reactants)
Given:
ΔG° for the hydrolysis of fructose-1-phosphate = -16.0 kJ/mol
ΔG° for the hydrolysis of ATP = -30.5 kJ/mol
The reaction we want to calculate the ΔG° for is:
fructose + ATP → fructose 1-phosphate + ADP
From the given information, we can break down the reactants and products:
Sum of reactants:
fructose + ATP
Sum of products:
fructose 1-phosphate + ADP
Now, we can calculate the ΔG° for the overall reaction:
ΔG° (overall reaction) = ΔG° (sum of products) - ΔG° (sum of reactants)
ΔG° (overall reaction) = (-16.0 kJ/mol) + (-30.5 kJ/mol)
ΔG° (overall reaction) = -46.5 kJ/mol
Therefore, the free energy change (ΔG°) for the reaction of fructose + ATP → fructose 1-phosphate + ADP is -46.5 kJ/mol.
2. Mr. Hamilton, a diabetic patient at your clinic, is diagnosed with ketoacidosis by the physician. The
physician explains that if a diabetic patient does not take the insulin that is normally prescribed and if
the resulting hyperglycemia is left untreated, it can lead to an emergency condition called ketoacidosis
This occurs when ketones, a group of acids that can build up in the bloodstream, accumulate, and
change the balance of acids in the blood. The patient's wife, Mrs. Hamilton, asks if you can explain in
basic terms why such a change is potentially life threatening.?
Answers
Ketoacidosis is a potentially life-threatening condition that happens when the body begins breaking down fat at very fast rate. The fat is converted by the liver and causes the blood to become acidic and needs to be treated immediately.
What is Diabetic Ketoacidosis (DKA)?Diabetes-related ketoacidosis (DKA) is a potentially fatal condition that affects diabetics. It happens when the body begins breaking down fat at an abnormally fast rate. The fat is converted by the liver into a fuel called ketones, which causes the blood to become acidic.
Diabetic ketoacidosis symptoms frequently appear suddenly, sometimes within 24 hours. High blood sugar levels and high ketone levels in urine are some of the first signs for DKA.
Learn more on Diabetic Ketoacidosis (DKA) here: https://brainly.com/question/28096487
#SPJ1
Electrons farther away from the nucleus in an atom have _______ compared to those closer to the nucleus.Question options:A) lower energyB) higher energyC) higher or lower energy, depending on the elementD) the same energy
Answers
Answer
B) higher energy
Explanation
The closer the electrons are to the nucleus, the lower the energy level. The farther the electrons are from the nucleus, the higher the energy level. In the lowest energy level, only one orbital exists that can carry a maximum of two electrons.
Therefore, electrons farther away from the nucleus in an atom have higher energy compared to those closer to the nucleus.
The correct answer is option B) higher energy
what volume of a 3M aqeuous solution of sodium phosphate will react with an excess amount of an aqueous solution of iron ll chloride to produce 25 g of precipitate?
Answers
11.7 mL is the volume required for a 3M aqueous solution to produce 25 grams of the precipitate.
Volume of solution = 3M
Mass of precipitate = 25g
The chemical balance equation between sodium phosphate and iron(II) chloride is:
\(2 Na_{3} PO_4(aq) + 3 FeCl_2(aq) -- > Fe_3(PO_4)_2(s) + 6 NaCl(aq)\)
To calculate the moles of Iron phosphate,
moles of \(Fe_3(PO_4)_2\) = mass of \(Fe_3(PO_4)_2\) / molar mass of \(Fe_3(PO_4)_2\)
The molar mass of \(Fe_3(PO_4)_2\) is calculated as:
\(Fe_3(PO_4)_2\) = 3 Fe atoms x atomic mass of Fe + 2 \((PO_4)\) groups x molar mass of \((PO_4)\)
\(Fe_3(PO_4)_2\) = 3 x (55.845 mol) + 2(2 x 31.0g) + (8 x 16.0 g/mol)
\(Fe_3(PO_4)_2\) = 357.696 g/mol
The moles \(Fe_3(PO_4)_2\) produced:
moles of \(Fe_3(PO_4)_2\) = 25 g / 357.696 g/mol
moles of \(Fe_3(PO_4)_2\) = 0.070 moles
moles of \(Na_3PO_4\) = 0.070 moles / 2
moles of \(Na_3PO_4\) = 0.035 moles
The volume of solution required for \(Na_3PO_4\) :
moles of \(Na_3PO_4\) = M x V
V = moles of \(Na_3PO_4\) / M
V = 0.035 moles / 3 M
V = 0.0117 L
V = 11.7 mL
Therefore, the volume required is approximately 11.7 mL.
To learn more about sodium phosphate
https://brainly.com/question/14292730
#SPJ1
Waste management is important for the environment. Waste can cause pollution and disease if it is
not handled properly. There are many different methods of waste management to help ease the
environmental pressures imposed by waste. Modern methods include recycling, composting,
incineration, sustainability, landfill, integration, waste-to-energy, and waste minimization. The
planet depends on a successful approach to waste disposal.
Landfills are waste disposal sites. They are often man-made depressions in the ground covered with a
lining designed to prevent any leakage of waste materials. However, landfills can leak through the base
resulting in negative impacts on the surrounding environments. What negative impact to the
environment is caused by the production of methane gas in landfills?
A
air pollution
B)
soil fertility
groundwater pollution
D)
air pollution, groundwater pollution, soil fertility

Answers
Answer:
air pollution, groundwater pollution, soil fertility
Answer:air pollution
Explanation:
its a gas, so it will mainly effect the air
Rough ER is connected to the nucleus membrane and to
Answers
Rough ER is connected to the nuclear membrane and is responsible for the synthesis and processing of proteins.
The rough endoplasmic reticulum (ER) is a network of membrane-bound sacs and tubules that is studded with ribosomes on its surface. These ribosomes are responsible for protein synthesis. The rough ER is connected to the nuclear membrane because it receives instructions for protein synthesis from the DNA in the nucleus. This connection allows for the efficient transfer of genetic information from the nucleus to the rough ER.
Once the ribosomes on the rough ER synthesize proteins, the rough ER is also involved in the processing and modification of these proteins. It helps in folding the newly synthesized proteins into their functional three-dimensional shapes and also adds various modifications such as glycosylation (the addition of sugar molecules) or signal sequences that target the proteins to specific locations within or outside the cell.
After processing, the proteins may be transported to other parts of the cell or exported to the cell membrane or extracellular space. The connection between the rough ER and the nuclear membrane ensures a coordinated flow of genetic information and protein synthesis, allowing the cell to efficiently carry out its functions.
for such more questions on proteins
https://brainly.com/question/30245761
#SPJ8
Problems:
Show all three steps of using a formula - show the formula, fill in the formula, 8 solve for the answer.
22) Calculate the energy of a photon of radiation with a frequency of 9.50 x 10¹³ Hz.
HINT:
You can make the formula parts larger by dragging them to the size that you prefer. DELETE THIS BOX AFTER READING!
HINT:
To make a superscript, cntrl / cmmd. (period)

Answers
The energy of a photon of radiation with a frequency of 9.50 × 10¹³ Hz is 6.3 × 10⁻²⁰ J.
The Energy that comes from electromagnetic radiation is quantized, It exists in the form of small energy packets known as photons. The energy of a photon is dependent on the frequency at which it is propagating from the source of radiation.
The energy of a photon is given by the equation:
E = hν
here, E is the energy of photon
h is Plancks's constant whose value is 6.626 × 10⁻³⁴ Js
ν is the frequency
According to the question,
given, frequency ν = 9.50 × 10¹³ Hz
therefore,
E = 6.626 × 10⁻³⁴ Js × 9.50 × 10¹³ Hz (1 Hz = 1s⁻¹)
E = 6.2947 × 10⁻²⁰ J
E≈ 6.3 × 10⁻²⁰ J
Thus, energy of the photon is 6.3 × 10⁻²⁰ J
To know more about energy of photon here
https://brainly.com/question/25746629
#SPJ4
3. A Wilkinson’s catalyst is widely used in the hydrogenation of alkenes. Show a catalytic cycle, including: i. chemical structure of the catalyst, with complete stereochemistry ii. molecular geometry of catalyst iii. type of reactions involved iv. the appropriate starting material, reagent and solvent v. major and minor end-products vi. all intermediates, for each reaction stated in (iii)
Answers
We can see here that the catalytic cycle for the hydrogenation of alkenes using Wilkinson's catalyst involves several steps.
What are the steps involved?Here's an overview of the catalytic cycle, including the necessary details:
i. Chemical structure of the catalyst:
Wilkinson's catalyst, also known as chloridotris(triphenylphosphine)rhodium(I), has the following chemical structure: [RhCl(PPh3)3]
ii. Molecular geometry of the catalyst:
The Wilkinson's catalyst has a trigonal bipyramidal geometry around the rhodium center. The three triphenylphosphine (PPh3) ligands occupy equatorial positions, while the chloride (Cl) ligand occupies an axial position.
iii. Type of reactions involved:
The catalytic cycle involves several reactions, including:
Oxidative addition: The rhodium center undergoes oxidative addition, reacting with molecular hydrogen (H2) to form a dihydride intermediate.Alkene coordination: The alkene substrate coordinates to the rhodium center, forming a π-complex.Hydrogenation: The coordinated alkene undergoes hydrogenation, resulting in the addition of hydrogen atoms to the double bond and formation of a metal-alkyl intermediate.Reoxidation: The metal-alkyl intermediate reacts with a hydrogen molecule to regenerate the rhodium dihydride species.iv. Starting material, reagent, and solvent:
The starting material is an alkene, and the reagent is Wilkinson's catalyst ([RhCl(PPh3)3]). The reaction is typically carried out in a suitable solvent, such as dichloromethane (CH2Cl2) or tetrahydrofuran (THF).
v. Major and minor end-products:
The major end-product of the hydrogenation reaction is the fully saturated alkane, resulting from the addition of hydrogen across the double bond. The minor end-product may include cis- or trans-configured alkanes if the original alkene substrate possesses geometric isomers.
vi. Intermediates:
The intermediates in the catalytic cycle include:
Rhodium dihydride complex: [RhH2(PPh3)3]Alkene-Rhodium π-complex: [Rh(η2-alkene)(PPh3)3]Metal-alkyl intermediate: [Rh(alkyl)(PPh3)3]These intermediates play a crucial role in facilitating the hydrogenation reaction and enabling the catalytic cycle to proceed.
Learn more about Wilkinson’s catalyst on https://brainly.com/question/31972308
#SPJ1