explain why the ph at the half-titration point is equal to the pka in your experiment
Answers
In an experiment, the pH at the half-titration point is equal to the pKa because the half-titration point is the point at which exactly half of the acid or base has been neutralized by the titrant.
At this point, the concentration of the acid and the conjugate base are equal, and they are in the process of forming equal amounts of H+ and A-.The pKa is defined as the negative logarithm of the acid dissociation constant (Ka), which is a measure of the strength of the acid. It represents the pH at which half of the acid is dissociated and half is undissociated. Therefore, at the half-titration point, the concentration of the acid and its conjugate base are equal, and the pH is equal to the pKa of the acid. For example, if the pKa of the acid being titrated is 4.5, then the pH at the half-titration point would be 4.5.
To learn more about conjugate base, click here:
https://brainly.com/question/30225100
#SPJ11
Related Questions
tính nồng độ của các ion trong dung dịch CA (OH)2 0.7M
Answers
Answer:
what are you saying ma guy?
Explanation:
Argon is a noble gas.
Explain why no product is formed when magnesium and argon are heated together.
[2 mark
Answers
Argon is a noble gas. argon no product is formed when magnesium and argon are heated together because argon is extremely stable.
The argon is a noble gas. noble gases are extremely stable. the noble gases have their octet complete and follow the octet rule. the outermost shell in the argon atom have the eight electron that eight valence electrons and it is fulfilling the octet rule. this means they have very little little tendency to gain or loss the electrons and they are very unreactive.
Thus, Argon is a noble gas. argon no product is formed when magnesium and argon are heated together because argon is extremely stable.
To learn more about argon here
https://brainly.com/question/3125326
#SPJ1
select all resonance structure of the radical produced when a hydrogen atom is abstracted from oh group
Answers
Resonating structures of BHT are shown below.
Except option lV all three are the resonating structures of BHT.
The electronic bonding of a single polyatomic species, including fractional bonds and fractional charges, is described by resonance structures, which are a collection of two or more Lewis structures.
When a molecule is represented by two or more hybrid structures, and those structures differ in the positions of electrons rather than atoms, the structures are referred to as resonance-producing structures.
It is a phenomena that occurs when the frequency of an externally applied periodic force equals the inherent frequency of a body, causing the body to start vibrating more vigorously.
To learn more about Resonating structures visit:https://brainly.com/question/29547999
#SPJ4

A homogeneous gas reaction A→3R has reported rate at 215∘c −γA=10−2C1/2 (mol/lit bee) Find the space time needed for 80% conversion of a 50%. A −50% inert feed to a plug flow reactor operating at 215∘. and 5 atm ( CAO0=0.0325 mol/lit ) using numerical integration method. (Minimize the pereentage of error in calculation).
Answers
By integrating the differential equation and determining the residence time at which the conversion reaches 80%, we can find the space time needed. The goal is to minimize the percentage of error in the calculation.
To solve this problem, we need to set up and solve the differential equation for the plug flow reactor. The rate equation given is γA = 10^(-2)C^(1/2) (mol/lit bee), where γ is the reaction rate constant and C is the concentration of A.
The differential equation for the plug flow reactor can be written as:
dCA/dV = -rA
Where CA is the concentration of A, V is the reactor volume, and rA is the rate of reaction. Since the reaction is homogeneous and follows the stoichiometry A → 3R, the rate of reaction is given by:
rA = -1/3 dCA/dt
Using the chain rule, we can rewrite the differential equation as:
dCA/dV = -1/3 dCA/dt dV/dt
The volume V is related to the reactor residence time τ (space time) by:
V = F₀τ
Where F₀ is the inlet molar flow rate. In this case, the feed consists of 50% A and 50% inert, so the inlet molar flow rate is 0.0325 mol/lit * 0.5 = 0.01625 mol/lit.
Now, we can substitute the expressions for V and dV/dt into the differential equation and rearrange it as:
(1/τ) dCA/dτ = -1/3 dCA/dt
To solve this differential equation numerically, we can use a method like the fourth-order Runge-Kutta method. We start with the initial condition CA = CA₀ at τ = 0 and integrate the differential equation until the conversion reaches 80% (CA = 0.0325 * 0.5 * 0.2 = 0.00325 mol/lit).
By varying the residence time τ and checking the conversion, we can determine the residence time at which the conversion is closest to 80%. This residence time will give us the space time needed for 80% conversion.
To minimize the percentage of error in the calculation, we can adjust the step size in the numerical integration method to achieve a desired level of accuracy. Smaller step sizes generally lead to more accurate results but require more computational effort.
By implementing the numerical integration method and adjusting the step size, we can find the space time needed for 80% conversion with minimized error.
Learn more about numerical integration methods here: brainly.com/question/28990411
#SPJ11
What is formed by the interaction of lighting with oxygen ?
Answers
Answer:
Ozone or, also known as O^3 is commonly formed by oxygen's interaction with lightning, and is what make the air smell so fresh after it rains. Hope this helps on your gradpoint Earth Atmosphere Posttest!
Explanation:
Which element does the "X" in
the isotope notation
represent?
14X
6
A. C
B. O
C. Si
D. N
Answers
The element whose isotope is represented as \(_{6}^{14}X\) is carbon -14. The atomic number of carbon is 6 and the mass number for the given isotope is 14 amu.
What are isotopes?Isotopes are atoms of same element with different mass numbers. Isotopes slightly changes in chemical and physical properties between each other.
Almost elements in the periodic table are having isotopes. However not all of them are stable. Some of them for heavy metals are radioactive isotopes and are not stable.
The isotope is represented by showing mass number in top of the symbol and atomic number at the bottom. The atomic number 6 corresponds to carbon. Hence, option A is correct. Carbon -14 isotope is given here.
To find more on isotopes, refer here:
https://brainly.com/question/27475737
#SPJ2
Harry stores toxic chemical waste in a large steel tank that has only 15% of its volume underground. Jena lives in the wilderness and has her own gas pump connected to a 1,500-gallon tank of gasoline buried ten feet underground near her garage. Lars keeps three large tanks filled with formaldehyde and battery acid in his basement. Which of these people are covered by federal regulations regarding USTs?
Answers
Answer:
Only Harry and Jena
Explanation:
Under federal regulations, an UST is any one or a combination of tanks such that the volume of an accumulation of regulated substances is 10% or more beneath the surface of the ground.
Any UST system holding a mixture of hazardous waste and other regulated substances are also are not covered by federal regulations regarding USTs.
Farm or residential tank of capacity more than 11 gallons used for storing motor fuel is covered by federal regulations regarding USTs.
According to the given question,
Only Harry and Jena are covered by federal regulations regarding USTs.
How do scientists use differences and similarities to identify life forms? I NEED TO KNOW ASAP
Answers
Hello!
Answer:
They utilize differences and similarities because some things look almost identical but like one or more things are different about another.
Explanation:
Like a lizard, two lizards have the same face, body shape, etc. but the markings are different, that allows scientists to identify the type of lizard the lizards are, because of the difference.
Hope this helps!
-Starr
5. Which substance represents a compound?
(1) C(s)
(2) Co(s)
(3) CO(g)
(4) O₂(g)
Answers
Calculate the pH of a 0.25 M solution of NaNO2 (Ka(HNO2) = 4.5 x 10^-4) (1.97)
a) pH = 3.35
b) pH = 4.45
c) pH = 5.55
d) pH = 6.65
Answers
The pH of a 0.25 M solution of NaNO2= 6.65.
Given the concentration of NaNO2, we can find the concentration of NaOH and HNO2 as follows:
NaNO2 = 0.25 MNaOH = HNO2 = x
(since they have equal concentrations due to the stoichiometry of the reaction)
Thus, we can write the equilibrium constant expression as:
Ka = x^2/0.25
Now, let's solve for x:
x^2 = 0.25 x 4.5 x 10^-4x = √(0.25 x 4.5 x 10^-4) = 0.015
This value represents the concentration of both HNO2 and NaOH. Since we are interested in pH, we need to find the concentration of H+ ions using the following equation:
Kw = [H+][OH-]
Since we have found the concentration of OH- (which is the same as the concentration of NaOH),
we can solve for H+:
Kw = 1.0 x 10^-14[H+][0.015] = 1.0 x 10^-14[H+] = 6.7 x 10^-13
Finally, we can find pH:
pH = -log[H+]pH = -log(6.7 x 10^-13)pH = 6.65
Therefore, the correct option is d) pH = 6.65.
learn more about pH here
https://brainly.com/question/172153
#SPJ11
For a solution, [H+] = 3 x 10-11M.
What is the pH?
Answers
Answer:
pH≈10.5
Explanation:
-log(3 x 10-11)≈10.5
when holden goes to visit phoebe, how does she react to his arrival? how are the two similar and how are they different?
Answers
Catcher in the Rye is a young-adult fiction by J. Salinger. It is a novel based on the coming of the age and literary realism. Phoebe is disappointed with Holden's vindication.
Holden Caulfield is the protagonist and the narrator of the story who is intelligent but is expelled from the school. Phoebe is Holden's sister, she is infuriated by Holden's dismissal.
She knows that their father would be angry because Holden is expelled from school. Holden tries to explain his situation to his sister and tells her about his minimum chance of joining the military school.
He explains to her how bad is his school Pencey and he dislikes that place. But Phoebe interrupts him by saying that he doesn't like anything or anywhere.
Her challenge makes him think about the one thing that he likes the most and he couldn't. It shows Phoebe's concern to stimulate Holden. She is seen sympathetic and angry with her brother for not growing up.
Holden describes them in loving, caring terms, demonstrating his genuine affection and respect for them. How does Holden characterize Phoebe? Holden describes Phoebe as wise, overly affectionate at times, and overly emotional for her age.
Also as smart, tiny, and a little noisy. Holden, Phoebe, and Allie are siblings in the novel "The Catcher in the Rye." Holden looks up to his younger brother and sister as idealized versions of himself. Holden describes Allie as the most intelligent and pleasant member of his family.
To know more about the Holden, refer: https://brainly.com/question/1344932
#SPJ4
For the reaction,
NH.'(aq) + H¿O() == NHglaq) + HyO^ (ag)
the equilibrium constant K is
O kb for NHy
O Ka, for NH3
O Ka, for NH4
O kb, for NH4
Answers
The given reaction is:NH3(aq) + H2O (l) ⇌ NH4+(aq) + OH-(aq)Now, the equilibrium constant K for this reaction is defined as K = [NH4+][OH-]/[NH3][H2O]
Let's find out the acid dissociation constant (Ka) for the given reaction using this formula:
Ka = [NH4+][OH-]/[NH3]
Since NH3 is a weak base, it reacts with water in an acidic solution to form NH4+ ions. Hence, it can be concluded that the reaction NH3 + H2O ⇌ NH4+ + OH- is actually a base dissociation reaction of NH4+.
Thus, the acid dissociation constant (Ka) for NH4+ is:
Ka = Kw/Kb,
where Kb is the base dissociation constant of NH3 and Kw is the ionization constant of water.
Kw = [H+][OH-]
= 1.0 x 10^-14 at 25°C (at 25°C, the product of H+ and OH- concentrations in water is always equal to 1.0 x 10^-14)At equilibrium, [NH3] and [H2O] are in excess. Therefore, they are taken as constant and their product is replaced by a constant Kc.
Kc = [NH4+][OH-]So,
Ka = Kw/Kb = [H+][OH-]/[NH4+][OH-]
= [H+]/[NH4+]Hence, Ka for NH4+
= [H+]/[NH4+].
Therefore, the correct answer is O Ka, for NH4.
To know more about NH3 visit:
https://brainly.com/question/15400042
#SPJ11
HELPPPP!!!! SCIENCE!!!!
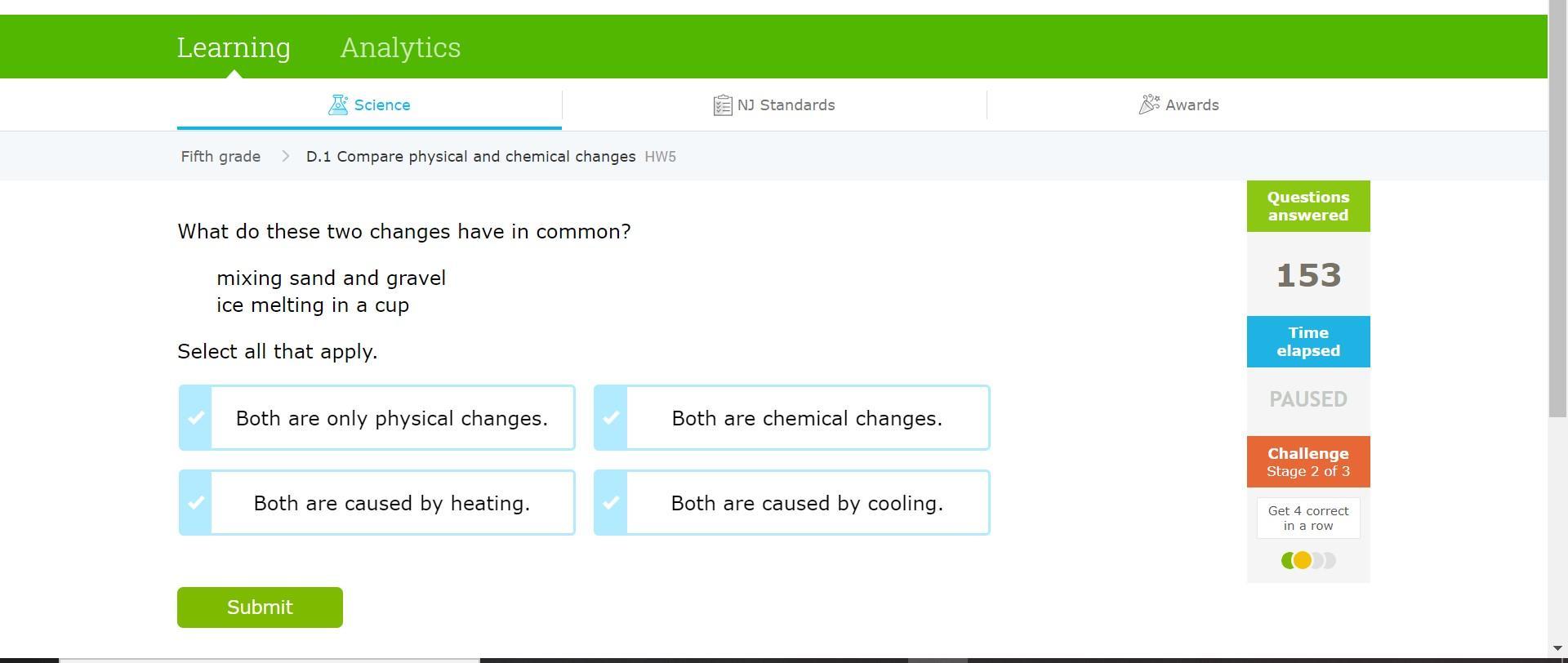
Answers
Answer:
Both are only physical changes
Which kind of sampling system uses a pump to draw air over a sorbent material?
Active
Passive
Dynamic
Constant-pressure
Answers
The type of sampling system that uses a pump to draw air over a sorbent material is an active sampling system.
Active sampling systems use a pump to draw air over a sorbent material in order to collect a sample. The pump creates a vacuum to draw air from the environment and then passes it over a sorbent material such as activated charcoal or silica gel. The sorbent material collects particles, gases and vapors from the air, which can then be analyzed. This type of system is ideal for measuring short-term, intermittent exposure to pollutants and is often used to monitor workplace air quality.
To learn more about silica gel click here https://brainly.com/question/30009290
#SPJ11
of a certain compound , known to be made of carbon and hydrogen, and to have a molecular molar mass of , is burned completely in excess oxygen, and the mass of the products carefully measured:
Answers
The compound X that have a Molecular molar mass of 138 g/mol is
Given here,
Compound x - 6g
malar mass of x - 138g/mol
Carbon dioxide - 13.39 g
water - 2.35g
So,
The moles of Water will be 0.13 mol
The moles of Hydrogen will be 0.26
Mass of hydrogen = 0.26 g
The moles of CO2 will be 0.304
The moles of carbon will be 0.304
Mass of Carbon = 3.65 g
Mass of Oxygen = Total mass of the compound - (mass of C + mass of H)
= 6.0 g - ( 3.65 + 0.262 ) g
= 2.09 g
Moles of oxygen = 0.131
Since Oxygen has the least moles, to find the molar ratio of the constituent atom, divide the moles of an atom from oxygen.
C = 0.304/0.131 = 2.8
C = 0.26/0.131 = 2
C = 0.131/0.131 = 1
convert 2.3 into a whole number
C = 2.3*3 = 7
H = 2*3 = 6
O = 1*3 = 3
Hence formula will be C7H6O3
What is molar mass?
The molar mass of a chemical compound is determined in chemistry by dividing the mass of a sample of that compound by the quantity of that compound, which is the number of moles in that sample, measured in moles. The molar mass of a substance is a bulk attribute rather than a molecular one. The compound's molar mass is an average over numerous samples, which frequently have different masses because of isotopes. A terrestrial average and a function of the relative abundance of the isotopes of the constituent atoms on Earth, the molar mass is most frequently calculated using the standard atomic weights. For converting between a substance's mass and amount in bulk amounts, the molar mass is the proper unit.
To know more about molar mass go to the given link:
https://brainly.com/question/837939
#SPJ4
Which sentence contains a common allusion?
O A. The meeting became extremely tense.
OB. The meeting turned into Waterloo.
O C. The meeting was held in Chicago.
OD. The meeting proceeded despite obstacles.
Answers
Answer:
B
Explanation:
The sentence that is a common allusion is:
B. The meeting turned into Waterloo.
All the other choices are not considered as an allusion
What is an allusion?It is an inferred or roundabout reference to an individual, occasion, or thing or to a piece of another text. Most references rely upon the comprehension that there is a gathering of data that is shared by the essayist and the peruser consequently. the peruser will get the author's referent.
The example of common allusion from the instances given are:
The meeting turned into Waterloo.
Find more information about Allusion here:
brainly.com/question/2427003
Le Chatelier's Principle governs what property?A. Reaction rateB. None of theseC. EquilibriumD. Catalysts
Answers
Equilibrium. Option C is correct
Explanations:What is Le Chatelier's principle?This law states that a new equilibrium state is achieved if the changes in temperature, pressure, concentration and volume will cause a predictable and opposing changes in the system.
This shows that Le Chatelier's principle can be used to predict the properties above to determine the effect equilibrium have on a system.
Based on the above explanations, we can conclude that Le Chatelier's Principle governs the property of Equilibrium.
Which of the following terms best describes the oxide of a metal?
Answers
Answer:
Zinc oxide is a metal oxide the term best describes the nature of zinc oxide is an amphoteric oxide.
Explanation:
Please Mark me brainlist
What is Difference Between Enantiomers and Diastereomers
Answers
Enantiomers and diastereomers are two types of stereoisomers, which are molecules that have the same molecular formula and connectivity. The key difference between enantiomers and diastereomers is their relationship to each other in terms of their symmetry and chirality.
Enantiomers are mirror images of each other, meaning they are non-superimposable and have the same physical and chemical properties except for the direction of rotation of plane-polarized light. Enantiomers have a single chiral center and are therefore isomers that differ only in the spatial orientation of their atoms around that chiral center. They have opposite configurations at every chiral center, so they have the same chemical and physical properties but different biological activities, pharmacological effects, and physiological properties. Diastereomers, on the other hand, are stereoisomers that are not mirror images of each other and have different physical and chemical properties. Diastereomers have multiple chiral centers and differ in their spatial orientation at some, but not all, of these centers. Diastereomers have different physical and chemical properties, and different biological activities, pharmacological effects, and physiological properties. In summary, enantiomers are mirror images of each other, have identical physical and chemical properties except for their interaction with plane-polarized light, and differ only in the configuration at one chiral center. Diastereomers, on the other hand, have different physical and chemical properties, and differ in their configuration at some, but not all, of their chiral centers.
Learn more about Enantiomers here:
https://brainly.com/question/21506956
#SPJ4
5.20763 to three significant figures
Answers
Answer:
5.21
Explanation: You can only have 3 digits, so you would round to the hundredths place
In science, a summary of observed behavior is referred to as a(n)
Answers
Answer:
Law
Explanation:
A law is a summary of observed (measurable) behavior, whereas a theory is an explanation of behavior. A law tells what happens; a theory (model) is our attempt to explain why it happens.
Draw a Lewis structure for SiF5-
Answers

Might be a bit messy but check the images.

what is the maximum current the lead-acid battery can supply for 75 h? the maximum current that can be supplied in 75 h is
Answers
The maximum current that a lead-acid battery can supply for 75 hours depends on its capacity, which is typically measured in ampere-hours (Ah). To determine the maximum current, it is necessary to know the battery's capacity.
The capacity of a lead-acid battery represents the total amount of charge it can store and deliver over a specific period. It is typically measured in ampere-hours (Ah). To calculate the maximum current that a lead-acid battery can supply for 75 hours, one needs to divide its capacity by the time duration.
For example, if the lead-acid battery has a capacity of 100 Ah, the maximum current it can supply for 75 hours would be obtained by dividing 100 Ah by 75 hours, resulting in a maximum current of approximately 1.33 amperes (A). The actual maximum current will vary based on the specific capacity of the battery in question.
Learn more about lead-acid battery here;
brainly.com/question/32156777
#SPJ11
Heat i removed from a 1. 6-microgram ample of liquid in a ealed container. The liquid tranform into a olid. After the tate change, what i the ma of the olid?
Answers
The mass of the solid after the state change would still be 1.6 micrograms. Mass is a measure of the amount of matter in an object, and it remains constant during a phase change (such as from a liquid to a solid) as long as no matter is added or removed from the system.
About MicrogramsIn the metric system, a microgram is a unit of mass equal to one million grams. The unit symbol is μg according to the International System of Units; the recommendation symbol in the United States and United Kingdom when conveying medical information is mcg.
Formula Micrograms :
1 kilogram
1 * 10^3 gram
1 microgram
1 * 10^-3 gram
1 kilogram / 1 microgram
10^3 / 10^-3
10^6
Learn More about Microgram at brainly.com/question/30460423
#SPJ4
When k–1 > k2 (that is, when the rate constant for dissociation of the enzyme substrate complex is greater than the rate constant for conversion to product), the KM is most analogous to
1) the Kd
2) the Ka
3) the Kcat
4) the 1/Kcat
Answers
When k1 > k2, the KM is most analogous to the Kd. This is because KM is the concentration of substrate at which the reaction rate is half of its maximum velocity, and Kd is the dissociation constant, which is the concentration of ligand at which half of the receptor binding sites are occupied.
In both cases, they represent the affinity of the enzyme or receptor for the substrate or ligand, respectively. The Ka is the association constant, which is the inverse of Kd, and is not directly related to KM. The Kcat is the turnover number, which represents the maximum number of substrate molecules converted to product per unit time by a single enzyme molecule when it is saturated with substrate, and 1/Kcat is the catalytic efficiency, which is not directly related to KM either.
When k-1 > k2 (the rate constant for dissociation of the enzyme-substrate complex is greater than the rate constant for conversion to product), the KM is most analogous to: 1) the Kd
Visit here to learn more about enzyme brainly.com/question/31385011
#SPJ11
an acid (ha) dissociates as follows: ha → h athe ph of the 0.80 m solution of ha is 1.38. what is the ka of ha? express your answer as a decimal, not an exponent.
Answers
The Ka of the weak acid HA is 0.0023
The pH of a solution of a weak acid is related to its dissociation constant (Ka) by the following equation:
pH = (1/2)(pKa - log [HA])
where HA represents the concentration of the weak acid, pKa is the negative logarithm of the dissociation constant, and log is the base 10 logarithm.
In this case, we are given that the concentration of the weak acid HA is 0.80 M, and the pH is 1.38. We can use the above equation to solve for Ka:
1.38 = (1/2)(pKa - log [HA])
2(1.38) = pKa - log (0.80)
2.76 + log (0.80) = pKa
pKa = 2.76 + (-0.0969) (taking the antilogarithm of both sides)
pKa = 2.663
Finally, we can use the relationship between Ka and pKa to calculate the Ka:
Ka = 10^(-pKa)
Ka = 10^(-2.663)
Ka = 0.0023
Click the below link, to learn more about pH of solution:
https://brainly.com/question/2288843
#SPJ11
WILL GIVE BRAINLIEST!! pls help
(04.05 MC)
Read the chemical equation shown.
3SO2 + 2HNO3 + 2H2O → 3H2SO4 + 2NO
Which statement is true about this chemical equation?
A) H (hydrogen) was oxidized and S (sulfur) was reduced.
B) H (hydrogen) was reduced and N (nitrogen) was oxidized.
C) S (sulfur) was oxidized and N (nitrogen) was reduced.
D) S (sulfur) was reduced and N (nitrogen) was oxidized.

Answers
Based on the chemical equation of the reaction, S (sulfur) was oxidized and N (nitrogen) was reduced.
The correct option is C.
What is a redox reaction?A redox reaction is a name for a reduction-oxidation reaction.
In a redox reaction, oxidation and reduction occur during the reaction.
Oxidation involves a loss of electrons and results in an increase in the oxidation number of the oxidized species.
Reduction involves a gain of electrons and results in a decrease in the oxidation number of the reduced species.
Considering the given equation of reaction:
3 SO₂ + 2 HNO₃ + 2 H₂O → 3 H₂SO₄ + 2 NO
The oxidation number of nitrogen changes from +5 in HNO₃to +2 in NO. Hence, it is reduced.
The oxidation number of sulfur changes from +4 tin SO₂ to +6 in H₂SO₄. Hence it is oxidized.
Learn more about redox reactions at: https://brainly.com/question/459488
#SPJ1Q4 This question relates the combustion reactions of acetylene, hydrogen and ethane. (a) Express the stoichiometric ecpigtions for the combustion reactions of acetylene, hydrogen and ethane with their respective standard heats of combustion obtained from physical property table. (b) Verify the standard heat of combustion of acetylene in Q4(a) by using heat of formation method. (c) The equation below shows the acerylene hydrogenation reaction: C2H2(g)+2H2(g)→C2H6(g) (i) Compute the standard heat of acetylcne hydrogenation reaction using tabulated heats of formation and heats of combustion. (ii) Verify the answer in Q4(e)(1) by using Hess's Law.
Answers
Stoichiometric equations for the combustion reactions ΔHf° (C2H2) = (2 x (-393.5)) + (-285.8) - (-1299.5) = +226.7 kJ mol-1(c) Acetylene hydrogenation reaction
Acetylene combustion reaction:C2H2 (g) + (5/2) O2 (g) → 2 CO2 (g) + H2O (l) ΔHc° = -1299.5 kJ mol-1 Hydrogen combustion reaction:2H2 (g) + O2 (g) → 2 H2O (l) ΔHc° = - 483.7 kJ mol-1Ethane combustion reaction:C2H6 (g) + (7/2) O2 (g) → 2 CO2 (g) + 3 H2O (l) ΔHc° = - 1560 kJ mol-1(b) Heat of formation method for verifying the standard heat of combustion of acetylene: The standard heat of combustion of acetylene from the heat of formation method is:ΔHc° (C2H2) = 2 ΔHf° (CO2) + ΔHf° (H2O) - 2 ΔHf° (C2H2) = -1299.5 kJ mol-1ΔHf° (CO2) = -393.5 kJ mol-1ΔHf° (H2O) = -285.8 kJ mol-1.
For verifying the answer in Q4(e)(1) using Hess's Law, we need to convert acetylene hydrogenation reaction into a combination of other reactions:Reaction 1:C2H2 (g) + (2.5) O2 (g) → 2 CO2 (g) + H2O (l) ΔH1 = -1299.5 kJ mol-1Reaction 2:2 CO2 (g) + 2.5 H2 (g) → C2H6 (g) + 5 O2 (g) ΔH2 = +1560 kJ mol-1After multiplying and adding the above equations, we get the required reaction as:C2H2 (g) + 2 H2 (g) → C2H6 (g) ΔH = -396.1 kJ mol-1.
To know more about reactions visit:
https://brainly.com/question/16737295
#SPJ11
I don’t really understand there’s questions

Answers
The molarity of the compound is 0.21M.
The pka of the acid is 3.98.
The miles or unknown acid is 5 × 10^-3
What is molar mass?Molar mass refers to the mass of one mole of a substance, which is usually expressed in grams per mole (g/mol). For example, the molar mass of water (H2O) is approximately 18 g/mol, which means that one mole of water weighs 18 grams.
On the other hand, pKa is a measure of the acidity of a substance. It is defined as the negative logarithm (base 10) of the acid dissociation constant (Ka). The pKa value reflects the strength of an acid, with lower values indicating stronger acids. For example, hydrochloric acid (HCl) has a pKa of approximately -6, while acetic acid (CH3COOH) has a pKa of approximately 4.76.
Learn more about molar mass on
https://brainly.com/question/837939
#SPJ1