Each of the structures for CIOCI, NCOH, and CINO has atoms connected in the order given in their formulas. Identify the type of hybridization for the central atom in each compound. Write the appropriate items to their respective bins.
a. nitrogen in CINO
b. oxygen in CIOCI
c. carbon in NCOH
1. sp
2 sp2
3. sp3
Answers
Answer:
see explanation
Explanation:
If we look at the structures of each of the compounds we can be able to deduce the accurate hybridization of each of the atoms listed.
Looking at ClNO - we can see that the nitrogen atom is linked to oxygen by a double bond and linked to chlorine by a single bond retaining a lone pair on nitrogen. Hence the nitrogen is in sp2 hybridized state.
For ClOCl, the compound has a sp3 hybridized oxygen atom linked to two chlorine atoms as expected retaining two lone pairs on oxygen.
For NCOH, the carbon atom is doubly bonded to oxygen hence it is sp2 hybridized.
Answer:
A) Label 2 (Sp2)
B) Label 3 (Sp3)
C) Label 1 (Sp)
Explanation:
A) Have 7 Lone Pairs (LP) to place. 1 on N. 1 becomes a double bond. Double bonds count as 1 Electron Group. Same for single bond and LP. Add these to get 3. 3 coresponds for SP2
B) 8LP. 2LP on O. 2 bonds to O. Add to get 4. 4-> Sp3
C) 5LP. C O bond is double. single bond to N. Add to get 2-> Sp
Related Questions
A solution containing 0.026 moles of H2O2 at 25.0 °C is placed in a coffee cup calorimeter and
allowed to decompose completely according to the thermochemical equation shown below. The final temperature of the solution is 44.9 °C. Calculate the enthalpy of the reaction shown, in kJ/mol. The mass of the solution is 30.0 g and the specific heat capacity of the solution
is 4.18 J/g°C.
CHEMICAL Formula is in the photo

Answers
The enthalpy (in kJ/mol) of the reaction, given that 0.026 moles of H₂O₂ at 25.0 °C is placed in a coffee cup calorimeter is 95.98 KJ/mol
How do i determine the enthalpy of the reaction?First, we shall determine the heat energy of the reaction. Details below:
Mass of solution = 30 gInitial temperature of statue (T₁) = 25 °CFinal temperature of statue(T₂) = 44.9 °CChange in temperature (ΔT) = 44.9 - 25 = 19.9 °C Specific heat capacity of solution (C) = 4.18 J/gºC Heat energy (Q) =?Q = MCΔT
Q = 30 × 4.18 × 19.9
Q = 2495.46 J
Finally, we shall determine the enthalpy of the reaction. Details below:
Heat absorbed (Q) = 2495.46 J = 2495.46 / 1000 = 2.49546 KJMole of H₂O₂ (n) = 0.026 moleEnthalpy of reaction (ΔH) =?Q = n × ΔH
2.49546 = 0.026 × ΔH
Divide both sides by 0.026
ΔH = 2.49546 / 0.026
ΔH = 95.98 KJ/mol
Thus, the enthalpy of reaction is 95.98 KJ/mol
Learn more about enthalpy change:
https://brainly.com/question/24170335
#SPJ1
7. What is the volume of the
composite
solid?
4 in.
3 in.
3 in.

Answers
Answer:
The volume of Component 1 is 36 cubic inches.
Explanation:
To calculate the volume of a composite solid, we need to determine the individual volumes of the different components and then add them together.
In this case, the composite solid consists of multiple components with the following dimensions:
Component 1:
Length: 4 inches
Width: 3 inches
Height: 3 inches
To find the volume of Component 1, we multiply the length, width, and height together:
Volume of Component 1 = Length x Width x Height = 4 in x 3 in x 3 in = 36 cubic inches
Therefore, the volume of Component 1 is 36 cubic inches.
Please provide the dimensions of the remaining components of the composite solid, and I will calculate the total volume by summing up the individual volumes.
Does sugar lose mass?
Can sugar melt?
Can sugar turn into water molecules?
Answers
Answer:
1. Yes
2. Yes
3. and for this one I would need to do some reasearch sorry
Explanation:
1. Sugar can lose mass but that depends on what you do to it
2. Sugar can melt It will melt into a clear liquid at 320 Fahrenheit
3. I don't know But I don't think it can FULLy turn into water molecules although it can mix with water molecules
Science 6
7. Which is NOT a heterogeneous mixture?
A. Fruit Salad
C. Marbles and Pebbles
B. Pencils and Pens
D. Vinegar
8. Why is a freshly squeezed orange juice called heterogeneous mixture?
A. No change can be observed.
B. The components were completely dissolved.
C. The components cannot be separated completely.

Answers
Answer:
7
ans: Fruit Salad
8
ans: The components were completely dissolved
which statement describes the components of a mixture
Answers
Answer:
Uh to answer the question we need to SEE the statements..
Determine the [H+], [OH−] , and pOH of a solution with a pH of 7.41 at 25 °C.
[H+]=
[OH−]=
pOH=
Answers
Answer:
[H+] = 2.96 x 10^(-9) M
[OH-] = 3.02 x 10^(-7) M
pOH = 6.59
Step-by-Step Explanation:
To determine the [H+], [OH-], and pOH of a solution with a pH of 7.41, we can use the relationship between pH, [H+], and pOH:
pH + pOH = 14
Given that the pH is 7.41, we can subtract it from 14 to find the pOH:
pOH = 14 - pH
pOH = 14 - 7.41
pOH = 6.59
Since pH + pOH = 14, we can also determine the [H+] and [OH-] using the pOH value:
pOH = -log[OH-]
6.59 = -log[OH-]
To solve for [OH-], we can take the antilog of both sides:
[OH-] = 10^(-pOH)
[OH-] = 10^(-6.59)
Using the relationship [H+][OH-] = 1 x 10^(-14) at 25 °C, we can determine [H+]:
[H+] = (1 x 10^(-14)) / [OH-]
[H+] = (1 x 10^(-14)) / (10^(-6.59))
Calculating the values:
[H+] = 2.96 x 10^(-9) M
[OH-] = 3.02 x 10^(-7) M
pOH = 6.59
Q: How does the starting
position affect the speed of a
ball rolling down a ramp?
I NEED HELPP PLSSS ASAP!!!
Answers
The force of gravity points straight down, but a ball rolling down a ramp doesn't go straight down, it follows the ramp. Therefore, only the component of the gravitational force which points along the direction of the ball's motion can accelerate the ball. The other component pushes the ball into the ramp, and the ramp pushes back, so there is no acceleration of the ball into the ramp. If the ramp is horizontal, then the ball does not accelerate, as gravity pushes the ball into the ramp and not along the surface of the ramp. If the ramp is vertical, the ball just drops with acceleration due to gravity. These arguments are changed a bit by the fact that the ball is rolling and not sliding, but that only affects the magnitude of the acceleration but not the fact that it increases with ramp steepness.
Andrew walks through his garden and observes that the shapes of dewdrops are not always the same. Suppose he wants to investigate using the scientific method. Which questions are testable questions that he can ask to look into the reasons for the different shapes? PLEASE HELP LOTS OF POINTS AND BRAILIEST
Answers
Answer:
A scientific method is the one which is independent of the person taking the measurements. Hence, a method which includes a scientific instrument/experiment, systematic observation, measurement, testing etc and is independent of personal feelings or opinions is known as a scientific method.
The measurement of temperature, testing of the material of surface, and moisture content in the atmosphere are testable questions and part of scientific method to find the reasons of different shapes of dewdrops. whether the different shapes are formed due to change in temperature, surface or moisture content in the atmosphere or not? The result of this measurement would not depend on the scientist doing the experiment. Scientific instruments would be used which would give the same reading. But the result of the observation of most unusual shape and pleasing shape of drop is purely dependent on one observer. It cannot be generalized reason
Explanation:
Answer:
brainliest pls pls pls
Explanation:
The average atomic mass for germanium is 72.631 amu. If germanium occurred as a mixture of the two isotopes below, what would be the percent abundance of 73Ge in the mixture?
mass of 70Ge = 69.924 amu
mass of 73Ge = 72.923 amu
Percent abundance of 73Ge in nature = ________ %
Answers
Answer:
dora needs help
Explanation:
Answer:
...........................
If 4.44 mol of C,H₁2 reacts with excess O₂, how many moles of CO₂ will be produced by the following combustion reaction?
C₂H2 +80₂6H₂O +5C0₂
moles of CO₂:
mol
Answers
A combustion reaction is a type of chemical reaction that involves the rapid combination of fuel (typically a hydrocarbon) with oxygen, resulting in the release of energy in the form of heat and light.
Combustion reactions are often characterized by the presence of a flame and the production of carbon dioxide (CO₂) and water (H₂O) as products.
In the given balanced combustion reaction:
C₂H₂ + 5O₂ → 4H₂O + 2CO₂
The stoichiometric ratio indicates that 1 mole of C₂H₂ reacts with 2 moles of CO₂ produced. Therefore, if 4.44 moles of C₂H₂ react, we can calculate the moles of CO₂ produced using the ratio:
Moles of CO₂ = (4.44 mol C₂H₂) × (2 mol CO₂ / 1 mol C₂H₂)
Moles of CO₂ = 8.88 mol
Therefore, 8.88 moles of CO₂ will be produced in the combustion reaction.
For more details regarding combustion reaction, visit:
https://brainly.com/question/14335621
#SPJ1
When molecules in a substance vibrate faster and faster, what happens to the temperature of the substance?
Answers
The temperature increases (gets warmer)
ExplanationsWhen the molecules in a substsnce vibrate faster ans faster, it shows that there is an increase the spaces between the molecules thereby leading to an increase in the kinetic enrgy of the molecule
The presence of this extra spaces causes the molecule to move freely thereby increasing the temperature of the substance.
In summary, the higher the vibration, the higher the kinetic energy, and the higher the temperature.
a layer of paint can be used to prevent iron rusting true or false
Answers
Answer:
true
Explanation:
This layer will prevent moisture from reaching the metal and therefore prevent rust. oil paint especially
What is a gas in the atmosphere that blocks high amount of infrared light?
What are these types of gases called?
Answers
The gases in the Earth's atmosphere that block a high amount of infrared light are called greenhouse gases.
These include carbon dioxide (CO₂), methane (CH₄), nitrous oxide (N₂O), and fluorinated gases, among others.
Greenhouse gases trap heat within the Earth's atmosphere and play a significant role in regulating the Earth's temperature.
However, when their concentration increases beyond natural levels, they can cause the Earth's temperature to rise, leading to global warming and climate change.
To know more about Earth's atmosphere, visit:
https://brainly.com/question/13154099
#SPJ1
c) Discuss precision and Accuracy as they relate to types of errors.
what is the answer
Answers
Precision relates to the consistency and reproducibility of measurements, while accuracy reflects how close measurements are to the true value.
Precision and accuracy are two important concepts in the context of errors in measurements. While they both pertain to the quality of data, they refer to different aspects.
Precision refers to the degree of consistency or reproducibility in a series of measurements. It reflects the scatter or spread of data points around the average value. If the measurements have low scatter and are tightly clustered, they are considered precise. On the other hand, if the measurements have a high scatter and are widely dispersed, they are considered imprecise.
Accuracy, on the other hand, refers to the closeness of measurements to the true or target value. It represents how well the measured values align with the actual value. Accuracy is achieved when measurements have a small systematic or constant error, which is the difference between the average measured value and the true value.
Errors in measurements can be classified into two types: random errors and systematic errors.
Random errors are associated with the inherent limitations of measurement instruments or fluctuations in the measurement process. They lead to imprecise data and affect the precision of measurements. Random errors can be reduced by repeating measurements and calculating the average to minimize the effect of individual errors.
Systematic errors, on the other hand, are caused by consistent biases or inaccuracies in the measurement process. They affect the accuracy of measurements and lead to a deviation from the true value. Systematic errors can arise from factors such as instrumental calibration issues, environmental conditions, or experimental techniques. These errors need to be identified and minimized to improve the accuracy of measurements.
In summary, precision refers to the degree of consistency or reproducibility of measurements, while accuracy refers to the closeness of measurements to the true value. Random errors affect precision, while systematic errors affect accuracy. To ensure high-quality measurements, both precision and accuracy need to be considered and appropriate techniques should be employed to minimize errors.
Know more about Precision here:
https://brainly.com/question/30461151
#SPJ8
A sample of air occupies 4.40 L when the pressure is 2.60 atm.
(a) What volume does the sample occupy at 6.20 atm at the same temperature?
L
(b) What pressure is required in order to compress it to 0.0290 L at the same temperature?
atm
Answers
Explanation:
Given that,
Initial volume, V₁ = 4.40 L
Initial pressure, P₁ = 2.6 atm
(a) Final pressure, P₂ = 6.2 atm
As per the relation,
\(P_1V_1=P_2V_2\\\\V_2=\dfrac{P_1V_1}{P_2}\\\\V_2=\dfrac{2.6\times 4.4}{6.2}\\\\V_2=1.84\ L\)
(b) Again using the above relation,
\(P_2=\dfrac{P_1V_1}{V_2}\\\\P_2=\dfrac{2.6\times 4.4}{0.0290 }\\\\P_2=394.48\ atm\)
Hence, this is the required ssolution.
Using the reading "Fossil Fuels" from lesson 12 describe the environmental and economic benefits and drawbacks of fossil fuels. Second, looking over the benefits and drawbacks, in your opinion, what do you think will happen to mining of fossil fuels in the next 50 years?
Answers
Fossil fuels are essential part of the power generation in the world. They are easily combustible and more reliable and cheaper. However, the burning of fossil fuels releases toxic gases to the environment.
What are fossil fuels ?Fossil fuels are fuel generated from the decomposition materials. Petroleum, coal, natural gas etc. are fossil fuels which are excavating from the earth.
Fossil fuels are non-renewable sources of energy. Hence, as the existing fossil sources are exhausted no more fossil fuel can be made. It is cheaper, reliable and easy to use.
However, the toxic hydrocarbon gases released from the burning of fossil fuels make the environment polluted. Therefore, overuse of fossil fuel definitely rise the atmospheric pollution.
Its use over the next 50 years, will increase the global warming and more of it will be exhausted.
Find more on fossil fuels :
https://brainly.com/question/3371055
#SPJ1
What does voltage describe?
Answers
The Voltage is the pressure from the electrical circuit of the power source that passes the current.
The Voltage is defined as the pressure from the electrical circuit of the power source that will passes the charged electrons that is the current through the conducting loop, it will enable them to do work because of the illuminating the light. The in simple terms is : voltage = pressure, and it is denoted as the volts and the symbol is the V.
The voltage is described as the force that causes the flow of the charged particles. The Voltage is also called as the electromotive force.
To learn more about voltage here
https://brainly.com/question/13177389
#SPJ1
What is the final temperature after 840 Joules is absorbed by 10.0g of water at 25.0
C?
Answers
The final temperature of the water is: T_final = 45.0°C
We can use the formula for the specific heat capacity of the water to solve this problem:
q = mcΔT
First, we can calculate the initial energy of the water:
q = mcΔT
q = (10.0 g) (4.184 J/g°C) (25.0°C)
q = 1,046 J
Next, we can calculate the final temperature after absorbing 840 J:
q = mcΔT
840 J = (10.0 g) (4.184 J/g°C) (ΔT)
ΔT = 20.0°C
Therefore, the final temperature of the water is:
T_final = T_initial + ΔT
T_final = 25.0°C + 20.0°C
T_final = 45.0°C
To know more about final temperature, here
brainly.com/question/11244611
#SPJ1
DEPARTMENT OF CHEMISTRY
UNIVERSITY OF LIBERIA
1. A 105.5mg sample of a white substance, suspected of being
cocaine (C17H21NO4), forms 2.79.3mg of CO2 and 66.46mg of
water on combustion. Chemical analysis shows that the
compound contains 4.68% N by mass. Would you conclude that
the white solid is cocaIne?
Answers
If 105.5mg sample of a white substance, suspected of being
cocaine (C17H21NO4), forms 2.79.3mg of CO2 and 66.46mg of
water on combustion, then the white solid is cocaine.
What is Combustion reaction?A Combustion reaction is defined as the chemical reaction which is generally a fuel that undergoes an oxidation by the reaction by the help of an oxidizing agent, which further results in the emission of energy.
It is mainly an exothermic process. It reaction between fuels and oxidents. Fuels such as wood and coal undergoes an endothermal pyrolysis. This result in the creation of fuel. Types of cocaine:Pure CocaineCrack CocaineSynthetic CocaineBlack CocaineThus, we concluded that the white solid is cocaine if If 105.5mg sample of a white substance, suspected of being
cocaine (C17H21NO4), forms 2.79.3mg of CO2 and 66.46mg of water on combustion.
learn more about Cocaine:
https://brainly.com/question/944545
#SPJ9
how are the different forms of energy converted
Answers
Answer:
Light energy can be converted to heat energy. Electrical energy can be converted to mechanical energy, light energy, heat energy, etc. Chemical energy can be converted to electrical energy. Thermal energy can be converted to heat energy
Explanation:
A constant electric current deposited 365 mg of Ag in 216 minutes from an aqueous Silver trioxonitrate (v). What is the Current?
Answers
The electric current is 0.025 A
Electric current refers back to the go with the flow of energy in an electronic circuit and to the amount of strength flowing through a circuit. it's far measured in amperes (A). the bigger the cost in amperes, the more energy is flowing within the circuit.
Ag+ + e¯ →Ag
1F deposits 107.87 g/mol (molecular mass) of silver
1F = 96500 C
Let, 107.87 g/mol needed = 96500 C
Number of coulombs required to deposit 0.3650 g of silver =(96500/107.87) 0.3650
Q = 326.5 C
According to Faraday’s law, Q = I x t
I = 326.5 C / (216 x 60 s) = 0.025 A
Learn more about electric current here:-https://brainly.com/question/2984202
#SPJ9
8. Jill conducted an experiment to investigate which plant food would make her
plants grow faster. She tried four different plant foods on plants of the same
type and age. Jill concluded that the plant that was fed Brand A grew the
fastest. Jill did not realize that the plant that was fed Brand A also received
more sunlight. (SC.7.N.1.5.3)
What error did Jill make in her experiment?
a) She used the wrong kind of plant.
b) She used too much plant food.
c) She did not control the variables.
Answers
In her experiment, Jill did not take into account the fact that the plant that was fed Brand A also received more sunlight. This means that there is another variable, besides the plant food, that could have affected the growth rate of the plant. By not controlling this variable, Jill's conclusion that Brand A was the best plant food may not be accurate. To properly control variables and make a valid conclusion, Jill should have ensured that all plants received the same amount of sunlight, or use other methods to control this variable as well.
Do air particles exert a force when they collide with objects
yes or no
Answers
Answer:
yes
Explanation:
Gas pressure is caused by the force exerted by gas molecules colliding with the surfaces of objects (Figure 5.2. 1). ... In fact, normal air pressure is strong enough to crush a metal container when not balanced by equal pressure from inside the container.
Answer: Yes Gas pressure is caused by the force exerted by gas molecules colliding with the surfaces of objects (Figure 5.2. ... In fact, normal air pressure is strong enough to crush a metal container when not balanced by equal pressure from inside the container
Explanation:
Please Help me solve for B
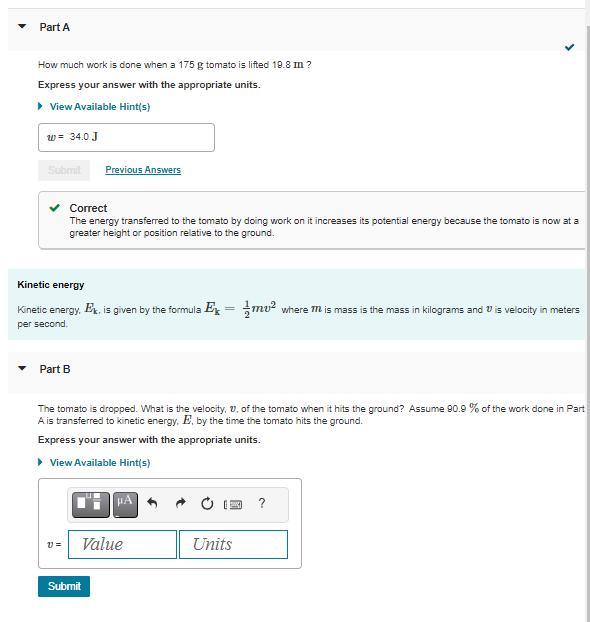
Answers
The velocity of the tomato when it hits the ground is approximately 13.49 meters per second.
The potential energy of the tomato is at the height of 10 meters. When the tomato hits the ground, most of the potential energy is E1 = 0.909*mgh.
By the conservation of energy principle, the kinetic energy \(E_1\) is equal to the kinetic energy \(E_2\) of the tomato just before it hits the ground.
The kinetic energy \(E_2\) is given by\(1/2mv^2\), where v is the velocity of the tomato just before it hits the ground. Equating \(E_1\) and \(E_2\) solving for v, we get:
\(v = \sqrt{(20.909gh)\)
Substituting the values of \(g = 9.81 m/s^2\)and h = 10 m, we get:
v = \(\sqrt{(20.9099.81*10)}\) = 13.49 m/s
To know more about potential energy, here
brainly.com/question/24284560
#SPJ1
--The complete Question is, Suppose a tomato is dropped from a height of 10 meters. If 90.9% of the work done on the tomato is converted to kinetic energy by the time it hits the ground, what is the velocity (in meters per second) of the tomato when it hits the ground? --
CuBr2 percent composition
Answers
The percent composition of CuBr₂ is approximately 28.46% of Cu and 71.54% of Br.
To determine the percent composition of CuBr₂ (copper(II) bromide), we need to calculate the mass of each element in the compound and then divide it by the molar mass of the entire compound.
The molar mass of CuBr₂ can be calculated by adding up the atomic masses of copper (Cu) and bromine (Br) in the compound. The atomic masses of Cu and Br are approximately 63.55 g/mol and 79.90 g/mol, respectively.
Molar mass of CuBr₂ = (63.55 g/mol) + 2(79.90 g/mol) = 223.35 g/mol
Now, let's calculate the percent composition of each element in CuBr₂:
Percent composition of copper (Cu):
Mass of Cu = (63.55 g/mol) / 223.35 g/mol × 100% ≈ 28.46%
Percent composition of bromine (Br):
Mass of Br = 2(79.90 g/mol) / 223.35 g/mol × 100% ≈ 71.54%
Therefore, the percent composition of CuBr₂ is approximately:
- Copper (Cu): 28.46%
- Bromine (Br): 71.54%
These values represent the relative mass percentages of copper and bromine in the compound CuBr₂.
for more such questions on composition
https://brainly.com/question/28250237
#SPJ8
Select the answer from the drop-down, ranging from true or false.
Mixtures have components that are always in fixed ratios.
[ true or false ]
Compounds can only be separated physically.
[ true or false ]
The components of a mixture keep their unique properties.
[ true or false ]
Mixtures can be physically separated
[ true or false ]
Mixtures are pure substances
[ true or false ]
Compounds are pure substances
[ true or false ]
Answers
Answer:
False, False, True, True, False, True
Explanation:
Mixtures have components that are always in fixed ratios. FALSE. The ratio of components in a mixture is variableCompounds can only be separated physically. FALSE. Compounds can only be separated chemically.The components of a mixture keep their unique properties. TRUE. Mixtures can be physically separated. TRUE. Mixtures are pure substances. FALSE. Mixtures are formed by a mixture of 2 or more pure substances, not bonded chemically.Compounds are pure substances. TRUE.true? no false. ... no its true uhhhhh no gotta be false true. false. true. fase. gotta be tru false or maybe its true? nah its false
pls brainliest?
What volume of CO2(g), measured at STP is produced if 15.2 grams of CaCO(s) is heated?
Answers
Answer:
Volume = 3.4 L
Explanation:
In order to calculate the volume of CO₂ produced when 15.2 g of CaCO₃ is heated, we need to first write out the balanced equation of the thermal decomposition of CaCO₃:
CaCO₃ (s) + [Heat] ⇒ CaO (s) + CO₂ (g)
Now, let's calculate the number of moles in 15.2 g CaCO₃:
mole no. = \(\mathrm{\frac{mass}{molar \ mass}}\)
= \(\frac{15.2}{40.1 + 12 + (16 \times 3)}\)
= 0.1518 moles
From the balanced equation above, we can see that the stoichiometric molar ratios of CaCO₃ and CO₂ are equal. Therefore, the number of moles of CO₂ produced is also 0.1518 moles.
Hence, from the formula for the number of moles of a gas, we can calculate the volume of CO₂:
mole no. = \(\mathrm{\frac{Volume \ in \ L}{22.4}}\)
⇒ \(0.1518 = \mathrm{\frac{Volume}{22.4}}\)
⇒ Volume = 0.1518 × 22.4
= 3.4 L
Therefore, if 15.2 g of CaCO₃ is heated, 3.4 L of CO₂ is produced at STP.
please help this is due today which ones are reactive and which arent?

Answers
Answer:
I' really don't know I'm sorry
HELP PLSSS
Why is the water cycle important for ecosystems?
A. The water cycle helps plants keep and store water. The cycle makes sure that water stays at earth's surface and is available to ecosystems.
B. The water cycle photosynthesis to make clouds. These clouds make surface water that animals drink to survive in there ecosystem.
C. The water cycle permanently removes water from the underground and from large bodies of water and transports it to terrestrial ecosystems
D. The water cycle helps recycle and transport water around the planet. The cycle replenishes the water supply of ecosystems.
Answers
Answer:
The water cycle is often taught as a simple circular cycle of evaporation, condensation, and precipitation. Although this can be a useful ...
Explanation:
Answer: Your answer will be D)
Explanation: Hope this helped
Which treatment(s) will help remove contaminants from minerals or from the pipes carrying water from a source? you can select more than one (Water Contamination Gizmos) **ONLY ANSWER IF YOU ACTUALLY KNOW ❗️❗️**
answer choices:
Sedimentation
Disinfection
Filtration
Coagulation

Answers
Sedimentation, filtration, and coagulation are the treatments that will help remove contaminants from minerals or from the pipes carrying water from a source.
Sedimentation is a process in which suspended particles settle out of water. It is one of the most basic techniques for removing particles from water. As particles settle, they become trapped in the bottom of a container or settle to the ground in an outdoor setting
Filtration is a method of removing particles from a fluid. It is a physical or chemical separation method that separates solids from fluids (liquids or gases) by adding a medium through which only the fluid can pass.
Coagulation is the process of using chemicals to remove contaminants from water. By creating a chemical reaction, coagulation destabilizes particles and causes them to clump together. This helps to remove the contaminants from the water.
Disinfection is the process of eliminating or destroying pathogens that cause infection. Disinfection eliminates harmful microorganisms by destroying or inactivating them. The disinfectant is a chemical or physical agent that is used to destroy or inactivate harmful microorganisms.
Know more about Filtration here:
https://brainly.com/question/29756050
#SPJ8